 Scheme1.
Monofluoromethylations with various electrophilic substrates
Scheme1.
Monofluoromethylations with various electrophilic substrates

银催化9-联烯嘌呤与氟代双(苯磺酰基)甲烷的单氟甲基化反应
-
关键词:
- 单氟甲基化反应
- / 9-联烯嘌呤
- / 氟代双(苯磺酰基)甲烷
English
Ag-Catalyzed Monofluoromethylation of Purin-9-yl Allenes with Fluorobis(phenylsulfonyl)methane
-
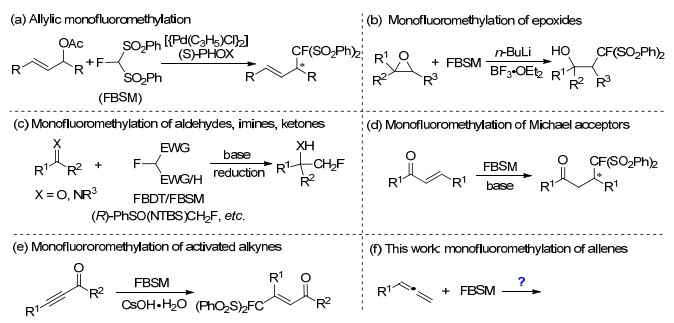
Monofluoromethylation has attracted substantial attention in organic synthesis, owing to the increasing important application of selectively fluorinated organics in medicinal chemistry and materials sciences.[1] Based on the significant pioneering studies by Shibata and Toru, [2] Hu, [3] Prakash and Olah, [4] fluorobis(phenylsulfonyl)methane (FBSM) has been demonstrated successfully as a fluoromethide equivalent. In 2006, Shibata and Toru et al.[2] reported the allylic monofluoromethylation of allyl acetates with FBSM to afford the fluorobis(phenylsulfonyl)methy-lated products in excellent results (Scheme 1a). Hu and coworkers[3] developed the monofluoromethylation of epoxides with FBSM to give the β-fluoroalkyl alcohols in high regioselectivity (Scheme 1b). Subsequently, several nucleophilic monofluoromethylation reagents including 2-fluoro-1, 3-benzodithiole-1, 1, 3, 3-tetraoxide (FBDT), [5a] TMSCF(SO2Ph)2, [5b] and (R)-PhSO(NTBS)CH2F[5c] were developed and reacted with various electrophilic substrates, including alcohols, [4] allylic carbonates, [6] aldehydes/imines/ketones (Scheme 1c), [5, 7] α, β-unsaturated ketones/aldehydes (Scheme 1d), [8] activated alkynes (Scheme 1e), [9] and others.[10] To the best of our knowledge, there has been no report on the monofluoromethylation of allenes (Scheme 1f). Thus, we wish to develop the monofluoromethylation of allenes with FBSM.
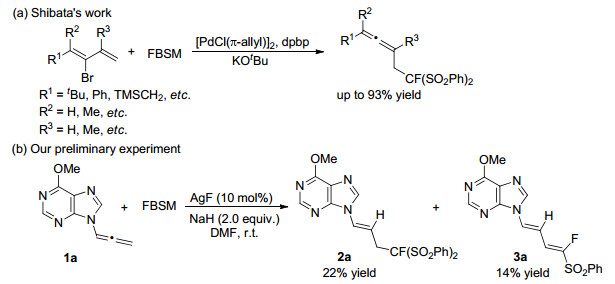
In 2009, Ogasawara and Shibata group[11] reported a Pd-catalyzed monofluoromethylation of substituted 2-bromo-1, 3-dienes with FBSM (Scheme 2a). Under these optimized conditions, the reactions took place very cleanly for a wide range of 2-bromo-1, 3-diene substrates, affording fluorinated allenes in excellent yields. Thus, these alkyl, aryl, or silylmethyl substituted fluorobis(phenylsulfonyl)-methyl allenes are stable and inert in the present of FBSM and Pd-catalyst. To our surprise, when purin-9-yl allene 1a was employed with AgF as the catalyst, the monofluoromethylation could happen, giving the fluorobis(phenylsulfonyl)methylated adduct 2a in 22% yield, along with the desulfonation product 3a in 14% yield. Considering acyclic nucleosides containing fluorine atom exhibit potential antivirus activities, [12] as the continuation of our work on selective modification of nucleosides, [13, 14] herein, we describe the monofluoromethylation of purin-9-yl allenes with FBSM to generate acyclic nucleoside analogues containing monofluoromethyl group in their side chain.
1 Results and discussion
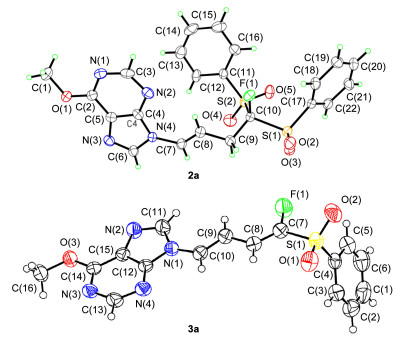
Initially, we studied the monofluoromethylation of FBSM to 6-methoxy-purin-9-yl allene (1a) catalyzed by AgF with NaH as the base in N, N-dimethylformamide (DMF) at room temperature for 12 h, giving the fluorinated products 2a and 3a in 22% and 14% yields, respectively (Table 1, Entry 1). The configuration of products 2a and 3a were confirmed by the singlecrystal X-ray diffraction analysis (Figure 1). It should be noted that the monofluoromethylation had an excellent chemoselectivity and the C=C bond in 2a has an exclusively E-configuration. Meanwhile, the two conjugated C=C bonds in the fluorinated 1, 3-butadiene 3a all have E-configuration. In order to decrease the desulfonation product 3a, several bases were evaluated, and the organic base Et3N was the best one, delivering 2a and 3a in 64% and 28% yields, respectively (Table 1, Entries 4~8). In addition, the amount of Et3N was screened and 1.5 equiv. of Et3N was suitable (Table 1, Entries 8~10). Other Ag salts were also explored, and no better results were observed (Table 1, Entries 11, 12). With Pd salts as catalysts, the monofluoromethylation did not occur (Table 1, Entries 13~15). Next, the catalyst loading was investigated, and 3 mol% was the best choice (Table 1, Entries 9 and 16, 17). When the reaction temperature was lowered to 0 ℃, only fluorinated 2a was obtained, although the yield was decreased to 59% yield (Table 1, Entries 16 and 18). To our delight, the fluorinated 2a could be obtained in 82% yield by prolonging the reaction time to 48 h (Table 1, Entry 19).

Entry Catalyst (mol%) Solvent Base (equiv.) Temp. Yieldb/% 2a 3a 1 AgF (10) DMF NaH (2) r.t. 22 14 2 AgF (10) CH2Cl2 NaH (2) r.t. N.R. N.R. 3 AgF (10) CH3CN NaH (2) r.t. 31 26 4 AgF (10) THF NaH (2) r.t. 53 43 5 AgF (10) THF Na2CO3 (2) r.t. 13 16 6 AgF (10) THF NaOtBu (2) r.t. Trace 41 7 AgF (10) THF iPr2EtN (2) r.t. 54 21 8 AgF (10) THF Et3N (2) r.t. 64 28 9 AgF (10) THF Et3N (1.5) r.t. 71 24 10 AgF (10) THF Et3N (1) r.t. 38 11 11 Ag2CO3 (10) THF Et3N (1.5) r.t. 34 11 12 AgSbF6 (10) THF Et3N (1.5) r.t. 42 17 13 Pd(dba)2 (10) THF Et3N (1.5) r.t. N.R. N.R. 14 Pd(OAc)2 (10) THF Et3N (1.5) r.t. N.R. N.R. 15 Pd(PPh3)4 (10) THF Et3N (1.5) r.t. N.R. N.R. 16 AgF (3) THF Et3N (1.5) r.t. 73 23 17 AgF (1) THF Et3N (1.5) r.t. 65 16 18 AgF (3) THF Et3N (1.5) 0 ℃ 59 0 19c AgF (3) THF Et3N (1.5) 0℃ 82 0 a Unless otherwise noted, the reaction conditions were: 1a (0.1 mmol), FBSM (1.1 equiv.), catalyst, solvent (1.0 mL), base for 12 h. b Isolated yield based on 1a. c Reaction time: 48 h, N.R.: no reaction. With the optimized reaction conditions established (Table 1, Entry 19), the substrate scope of purin-9-yl allenes was investigated (Table 2). When 6-ethoxy-purin-9-yl (1b) was used, the desired fluorobis(phenylsulfonyl)methylated adduct 2b was obtained in 86% yield. Next, several purin-9-yl allenes 1c~1g with amine substituents at C(6) position of purine fragment participated well in the monofluormethylation with FBSM, affording the corresponding adducts 2c~2g in 82%~86% yields. In the case of 6-phenyl-purin-yl allene (1h), the reaction proceeded well, giving the adduct 2h in 83% yield. In addition, when purin-9-yl allenes 1i~1j with amino or alkoxy group at the C(2) position of purine part were examined, the fluorobis(phenylsulfonyl)methylated adducts 2i~2j were generated in 77%~86% yields. It should be noted that the corresponding desulfonation products 3 could not be observed for all the examined purin-9-yl allenes.



a Reaction conditions: 1a~1j(0.2 mmol), FBSM (1.1 equiv.), AgF (3 mol%), Et3N (1.5 equiv.), THF (2.0 mL) at 0℃ for 48 h.bIsolated yield. Considering that the fluorobis(phenylsulfonyl)meth-ylated adduct 2a and fluorinated 1, 3-butadiene 3a could be formed simultaneously (Table 1, Entry 1), we wonder whether fluorinated 1, 3-butadiene 3a was generated from monofluoroalkene 2a. Thus, as shown in Eq. 1, the monofluoroalkene 2a was treated with excess of NaH in THF at room temperature for 12 h, and the fluorinated 1, 3-buta-diene 3a was obtained in quantitative yield. Owing to the stability of the conjugated 1, 3-butadiene in 3a, the elimination reaction of one molecular of phenylsulfonyl group in 2a was easy to happen.
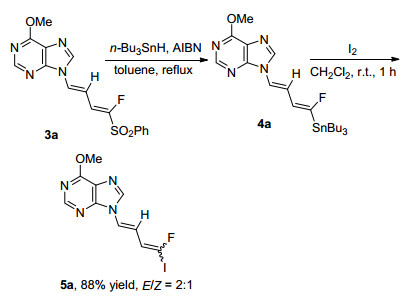
Subsequently, the desulfonation of fluorinated 1, 3-butadiene 3a was carried out (Scheme 3). In the presence of nBuSnH/AIBN, the phenylsulfonyl group in 3a could be efficiently transformed into a tributylstannyl group in 4a. Without isolation, organotin compound 4a could be converted into the iodinated product 5a in 88% yield within 2 steps under the conditions of I2 in CH2Cl2 at room temperature. As for iodinated 1, 3-butadiene 5a, the E/Z selectivity is only 2: 1, which was confirmed by 1H NMR analysis.
2 Conclusions
In conclusion, the monofluoromethylation of purin-9-yl allenes with FBSM has been achieved. The fluorobis-(phenylsulfonyl)methylated adducts could be obtained in excellent yields. In the presence of 3 mol% AgF, a series of purin-9-yl allenes with different substituents at C(6) and C(2) positions participated well in the monofluoromethylations, affording the corresponding monofluoroalkenes in 77%~86% yields. Meanwhile, the monofluoromethylation exhibited high chemoselectivities and E-selectivies. Furthermore, this monofluoromethylation of purin-9-yl allenes with FBSM provided a useful route to synthesize fluorinated acyclic nucleoside analogues.
3 Experimental section
3.1 Instrument and reagent
1H NMR spectra were recorded on commercial instruments (400 MHz). 13C NMR data were collected on commercial instruments (100 or 150 MHz) with complete proton decoupling. 19F NMR chemical shifts were determined relative to CFCl3 at δ 0.0. HRMS was recorded on a commercial apparatus (ESI Source). Melting points were reported without correction.
3.2 General procedure for monofluoromethylation
To a test tube, 6-methoxy-purin-9-yl allene (1a, 0.1 mmol, 18.8 mg) and AgF (3 mol%, 0.4 mg) were added, and tetrahydrofuran (THF, 1.0 mL) was added subsequently. The reaction was stirred at 0 ℃ for 0.5 h. Then, FBSM (1.1 equiv., 34.5 mg) and Et3N (1.5 equiv., 20.8 μL) were added subsequently. The reaction mixture was stirred at 0 ℃ for 48 h.
Work up procedure: Upon consumption of 1a, the mixture was filtered through Celite and the filtrate was concentrated to dryness. The crude adducts were then purified by flash column chromatography [eluent: V(ethyl acetate): V(petroleum ether)=1: 2] to afford the 3a in 82% yield.
3.3 Product structure characterization
(E)-9-(4-Fluoro-4, 4-bis(phenylsulfonyl)but-1-en-1-yl)-6-methoxy-9H-purine (2a): White solid. m.p. 165~166 ℃; 1H NMR (CDCl3, 400 MHz) δ: 8.55 (s, 1H), 8.03 (s, 1H), 7.92 (d, J=8.4 Hz, 4H), 7.70 (t, J=7.6 Hz, 2H), 7.54 (t, J=7.6 Hz, 4H), 7.05 (d, J=14.4 Hz, 1H), 6.41~6.34 (m, 1H), 4.20 (s, 3H), 3.36 (dd, J=17.6, 7.6 Hz, 2H); 13C NMR (CDCl3, 100 MHz) δ: 161.8, 159.8, 152.8, 136.4, 135.5, 135.3, 131.0, 129.3 (d, J=11.6 Hz), 125.5, 115.8 (d, J=7.0 Hz), 108.6 (d, J=6.0 Hz), 54.1, 31.5 (d, J=18.9 Hz); 19F NMR (CDCl3, 376 MHz) δ: -142.3; HRMS calcd for C22H19FN4NaO5S2 [M+Na+] 525.0673, found 525.0673.
(E)-6-Ethoxy-9-(4-fluoro-4, 4-bis(phenylsulfonyl)but-1-en-1-yl)-9H-purine (2b): White solid. m.p. 117~118 ℃; 1H NMR (CDCl3, 400 MHz) δ: 8.52 (s, 1H), 8.01 (s, 1H), 7.92 (d, J=7.6 Hz, 4H), 7.69 (t, J=7.6 Hz, 2H), 7.53 (t, J=7.6 Hz, 4H), 7.04 (d, J=14.4 Hz, 1H), 6.39~6.32 (m, 1H), 4.66 (q, J=7.2 Hz, 2H), 3.36 (dd, J=17.2, 7.2 Hz, 2H), 1.51 (t, J=6.8 Hz, 3H); 13C NMR (CDCl3, 100 MHz) δ: 161.0, 152.9, 151.0, 139.4, 135.6, 135.1, 131.0, 129.3, 125.2, 121.8, 110.5 (d, J=6.5 Hz), 63.5, 31.6 (d, J=19.3 Hz), 14.6; 19F NMR (CDCl3, 376 MHz) δ: -142.0; HRMS calcd for C23H22FN4O5S2 [M+H+] 517.1010, found 517.1008.
(E)-9-(4-Fluoro-4, 4-bis(phenylsulfonyl)but-1-en-1-yl)-N, N-dimethyl-9H-purin-6-amine (2c): White solid. m.p. 195~196 ℃; 1H NMR (CDCl3, 400 MHz) δ: 8.32 (s, 1H), 7.94 (d, J=8.4 Hz, 4H), 7.82 (s, 1H), 7.70 (t, J=7.2 Hz, 2H), 7.55 (t, J=7.6 Hz, 4H), 7.01 (d, J=14.4 Hz, 1H), 6.22~6.15 (m, 1H), 3.70~3.40 (br, 6H), 3.33 (ddd, J=18.0, 7.2, 1.2 Hz); 13C NMR (CDCl3, 100 MHz) δ: 154.9, 152.9, 149.6, 135.5, 135.2, 135.1, 130.9, 129.2, 125.4, 120.2, 114.3 (d, J=266.6 Hz), 109.0 (d, J=6.2 Hz), 38.6, 31.6 (d, J=18.9 Hz); 19F NMR (CDCl3, 376 MHz) δ:-142.2; HRMS calcd for C23H22FN5NaO4S2 [M+Na+] 538.0989, found 538.0985.
(E)-9-(4-Fluoro-4, 4-bis(phenylsulfonyl)but-1-en-1-yl)-6-(pyrrolidin-1-yl)-9H-purine (2d): White solid. m.p. 162~163 ℃; 1H NMR (CDCl3, 400 MHz) δ: 8.28 (s, 1H), 7.93 (d, J=8.0 Hz, 4H), 7.78 (s, 1H), 7.65 (t, J=7.2 Hz, 2H), 7.50 (t, J=7.6 Hz, 4H), 6.96 (d, J=14.4 Hz, 1H), 6.19~6.12 (m, 1H), 4.09 (s, 2H), 3.70 (s, 2H), 3.30 (dd, J=17.6, 7.6 Hz, 2H), 1.99 (d, J=22.4 Hz, 4H); 13C NMR (CDCl3, 100 MHz) δ: 161.0, 152.9, 151.0, 139.4, 135.6, 135.1, 131.0, 129.3, 125.2, 121.8, 115.6, 110.5 (d, J=6.7 Hz), 63.5, 31.6 (d, J=19.2 Hz), 14.6; 19F NMR (CDCl3, 376 MHz) δ: -142.2; HRMS calcd for C25H25FN5O4S2 [M+H+] 542.1327, found 542.1324.
(E)-9-(4-Fluoro-4, 4-bis(phenylsulfonyl)but-1-en-1-yl)-6-(piperidin-1-yl)-9H-purine (2e): White solid. m.p. 144~145 ℃; 1H NMR (CDCl3, 400 MHz) δ: 8.30 (s, 1H), 7.92 (d, J=7.6 Hz, 4H), 7.81 (s, 1H), 7.69 (t, J=7.6 Hz, 2H), 7.54 (t, J=7.6 Hz, 4H), 6.99 (d, J=14.8 Hz, 1H), 6.21~6.13 (m, 1H), 4.40~4.02 (br, 4H), 3.32 (dd, J=18.0, 7.6 Hz, 2H), 1.80~1.62 (m, 6H); 13C NMR (CDCl3, 100 MHz) δ: 153.8, 153.0, 149.9, 135.6, 135.2, 135.0, 131.0, 129.3, 125.4, 119.9, 114.2 (d, J=273.4 Hz), 109.1, 109.0, 46.5, 31.7 (d, J=19.1 Hz), 26.2, 24.9; 19F NMR (CDCl3, 376 MHz) δ: -142.1; HRMS calcd for C26H27FN5O4S2 [M+H+] 556.1483, found 556.1480.
(E)-4-(9-(4-Fluoro-4, 4-bis(phenylsulfonyl)but-1-en-1-yl)-9H-purin-6-yl)morpholine (2f): White solid. m.p. 157~158 ℃; 1H NMR (CDCl3, 400 MHz) δ: 8.30 (s, 1H), 7.90 (d, J=7.6 Hz, 4H), 7.82 (s, 1H), 7.67 (t, J=7.6 Hz, 2H), 7.51 (t, J=7.6 Hz, 4H), 6.99 (d, J=14.4 Hz, 1H), 6.26~6.18 (m, 1H), 4.46~4.06 (br, 4H), 3.80 (t, J=4.4 Hz, 4H), 3.31 (dd, J=17.6, 7.2 Hz, 2H); 13C NMR (CDCl3, 100 MHz) δ: 153.8, 152.8, 150.0, 135.6, 135.5, 135.0, 130.9, 129.2, 125.2, 120.0, 114.6 (d, J=266.7 Hz), 109.4 (d, J=6.5 Hz), 67.0, 45.5, 31.6 (d, J=19.0 Hz); 19F NMR (CDCl3, 376 MHz) δ: -141.9; HRMS calcd for C25H25FN5O5S2 [M+H+] 558.1276, found 558.1273.
(E)-9-(4-Fluoro-4, 4-bis(phenylsulfonyl)but-1-en-1-yl)-N, N-dibutyloxy-carbony-9H-purin-6-amine (2g): White solid. m.p. 174~175 ℃; 1H NMR (CDCl3, 400 MHz) δ: 8.86 (s, 1H), 8.18 (s, 1H) 7.92 (d, J=8.4 Hz, 4H), 7.69 (d, J=7.2 Hz, 2H), 7.53 (t, J=7.6 Hz, 4H), 7.08 (d, J=14.4 Hz, 1H), 6.50~6.42 (m, 1H), 3.38 (ddd, J=17.2, 7.6, 0.8 Hz, 2H), 1.45 (s, 18H); 13C NMR (CDCl3, 100 MHz) δ: 152.8, 152.2, 150.8, 150.4, 142.1, 135.6, 135.1, 131.0, 129.3, 129.2, 124.9, 111.3 (d, J=6.5 Hz), 84.1, 31.6 (d, J=19.2 Hz), 27.9; 19F NMR (CDCl3, 376 MHz) δ: -141.7; HRMS calcd for C31H35FN5O8S2 [M+H+] 688.1906, found 688.1902.
(E)-9-(4-Fluoro-4, 4-bis(phenylsulfonyl)but-1-en-1-yl)-6-phenyl-9H-purine (2h): White solid. m.p. 165~166 ℃; 1H NMR (CDCl3, 400 MHz) δ: 9.03 (s, 1H), 8.77 (d, J=7.2 Hz, 2H), 8.22 (s, 1H), 7.94 (d, J=7.6 Hz, 4H), 7.71 (t, J=7.2 Hz, 2H), 7.57~7.53 (m, 7H), 7.13 (d, J=14.8 Hz, 1H), 6.48~6.41 (m, 1H), 3.41 (dd, J=17.6, 7.2 Hz, 2H); 13C NMR (CDCl3, 100 MHz) δ: 155.5, 153.1, 151.5, 141.6, 135.6, 135.5, 135.2, 131.4, 131.3, 131.0, 130.0, 129.3, 128.9, 114.2 (d, J=266.4 Hz), 110.8 (d, J=6.5 Hz), 31.8 (d, J=19.1 Hz), 29.8; 19F NMR (CDCl3, 376 MHz) δ: -141.9; HRMS calcd for C27H22FN4O4S2 [M+H+] 549.1061, found 549.1060.
(E)-9-(4-Fluoro-4, 4-bis(phenylsulfonyl)but-1-en-1-yl)-6-methoxy-9H-purin-2-amine (2i): White solid, m.p. 204~206 ℃; 1H NMR (CDCl3, 400 MHz) δ: 7.92 (d, J=7.6 Hz, 4H), 7.68 (t, J=7.2 Hz, 2H), 7.63 (s, 1H), 7.52 (t, J=7.2 Hz, 4H), 6.86 (d, J=14.4 Hz, 1H), 6.15~6.08 (m, 1H), 4.94 (s, 2H), 4.07 (s, 3H), 3.34 (dd, J=18.0, 7.6 Hz, 2H); 13C NMR (CDCl3, 100 MHz) δ: 161.8, 159.8, 152.8, 136.4, 135.5, 135.3, 131.0, 129.2, 125.5, 115.9, 108.6 (d, J=6.2 Hz), 54.1, 31.5 (d, J=18.9 Hz); 19F NMR (CDCl3, 376 MHz) δ: -142.3; HRMS calcd for C22H20FN5NaO5S2 [M+Na+] 540.0782, found 540.0780.
(E)-9-(4-Fluoro-4, 4-bis(phenylsulfonyl)but-1-en-1-yl)-2, 6-dimethoxy-9H-purine (2j): White solid. m.p. 156~157 ℃; 1H NMR (CDCl3, 400 MHz) δ: 7.91 (d, J=7.6 Hz, 4H), 7.80 (s, 1H), 7.69 (t, J=7.2 Hz, 2H), 7.52 (t, J=7.6 Hz, 4H), 6.95 (d, J=14.4 Hz, 1H), 6.47~6.40 (m, 1H), 4.16 (s, 3H), 4.04 (s, 3H), 3.36 (dd, J=17.2, 7.6 Hz, 2H); 13C NMR (CDCl3, 100 MHz) δ: 162.3, 152.4, 138.1, 135.6, 135.5, 135.2, 131.0, 131.0, 129.2, 125.4, 117.8, 109.7 (d, J=6.6 Hz), 55.4, 54.6, 31.7 (d, J=19.2 Hz); 19F NMR (CDCl3, 376 MHz) δ: -141.7; HRMS calcd for C23H21FN4NaO6S2 [M+Na+] 555.0779, found 555.0779.
9-(-4-Fluoro-4-iodobuta-1, 3-dien-1-yl)-6-methoxy-9H-purine (5a): 1H NMR (CDCl3, 400 MHz) δ: 8.58 (s, 1H), 8.13 (s, 1H), 7.32~7.13 (m, 2H), 6.06 (dd, J=31.6, 10.4 Hz, 1H); 13C NMR (CDCl3, 100 MHz) δ: 161.3, 153.0, 139.4, 123.2, 121.2, 119.9, 117.1, 112.8, 105.3 (d, J=340 Hz), 54.5; 19F NMR (CDCl3, 376 MHz) δ: -62.2, -65.5.
Supporting Information Experimental procedures, the synthesis method of the starting materials, and compound characterization data (PDF), X-ray data for compounds 2a and 3a (CIF). The Supporting Information is available free of charge via the Internet at http://sioc-journal.cn/.
-
-
[1]
(a) Smart, B. E. J. Fluorine Chem. 2001, 109, 3.
(b) Kirsch, P. Modern Fluoroorganic Chemistry, Wiley-VCH, Weinheim, Germany, 2004.
(c) Kirk, K. L. J. Fluorine Chem. 2006, 127, 1013.
(d) Li, Y.; Ni, C.; Liu, J.; Zhang, L.; Zheng, J.; Zhu, L.; Hu, J. Org. Lett. 2006, 8, 1693.
(e) Bégué, J.-P.; Bonnet-Delpon, D. J. Fluorine Chem. 2006, 127, 992.
(f) Prakash, G. K. S.; Hu, J. Acc. Chem. Res. 2007, 40, 921.
(g) Müller, K.; Faeh, C.; Diederich, F. Science 2007, 317, 1881.
(h) Purser, S.; Moore, P. R.; Swallow, S.; Gouverneur, V. Chem. Soc. Rev. 2008, 37, 320.
(i) Hu, J.; Zhang, W.; Wang, F. Chem. Commun. 2009, 7465.
(j) Liao, F.-M.; Yu, J.-S.; Zhou, J. Chin. J. Org. Chem. 2017, 37, 2175(in Chinese).(廖富民, 余金生, 周剑, 有机化学, 2017, 37, 2175.)
(k) Liao, F.-M.; Cao, Z.-Y.; Yu, J.-S.; Zhou, J. Angew. Chem., Int. Ed. 2017, 56, 2459. -
[2]
Fukuzumi, T.; Shibata, N.; Sugiura, M.; Yasui, H.; Nakamura, S.; Toru, T. Angew. Chem., Int. Ed. 2006, 45, 4973 doi: 10.1002/(ISSN)1521-3773
-
[3]
Ni, C.; Li, Y.; Hu, J. J. Org. Chem. 2006, 71, 6829. doi: 10.1021/jo060955l
-
[4]
Prakash, G. K. S.; Chacko, S.; Alconcel, S.; Stewart, T.; Mathew, T.; Olah, G. A. Angew. Chem., Int. Ed. 2007, 46, 4933. doi: 10.1002/(ISSN)1521-3773
-
[5]
(a) Furukawa, T. ; Goto, Y. ; Kawazoe, J. ; Tokunaga, E. ; Naka-mura, S. ; Yang, Y. ; Du, H. ; Kakehi, A. ; Shiro, M. ; Shibata, N. Angew. Chem., Int. Ed. 2010, 49, 1642.
(b) Prakash, G. K. S. ; Shao, N. ; Zhang, Z. ; Ni, C. ; Wang, F. ; Haiges, R. ; Olah, G. A. J. Fluorine Chem. 2012, 133, 27.
(c) Shen, X. ; Miao, W. ; Ni, C. ; Hu, J. Angew. Chem., Int. Ed. 2014, 53, 775. -
[6]
(a) Liu, W. -B. ; Zheng, S. -C. ; He, H. ; Zhao, X. -M. ; Dai, L. -X. ; You, S. -L. Chem. Commun. 2009, 6604.
(b) Furukawa, T. ; Kawazoe, J. ; Zhang, W. ; Nishimine, T. ; Tokunaga, E. ; Matsumoto, T. ; Shiro, M. ; Shibata, N. Angew. Chem., Int. Ed. 2011, 50, 9684.
(c) Yang, W. ; Wei, X. ; Pan, Y. ; Lee, R. ; Zhu, B. ; Liu, H. ; Yan, L. ; Huang, K. -W. ; Jiang, Z. ; Tan, C. -H. Chem. Eur. J. 2011, 17, 8066. -
[7]
(a) Shen, X. ; Zhang, L. ; Zhao, Y. ; Zhu, L. ; Li, G. ; Hu, J. Angew. Chem., Int. Ed. 2011, 50, 2588.
(b) Ma, H. ; Matsuzaki, K. ; Yang, Y. -D. ; Tokunaga, E. ; Nakane, D. ; Ozawa, T. ; Masuda, H. ; Shibata, N. Chem. Commun. 2013, 49, 11206.
(c) Shen, X. ; Ni, C. ; Hu, J. Chin. J. Chem. 2013, 31, 878.
(d) Mizuta, S. ; Shibata, N. ; Goto, Y. ; Furukawa, T. ; Nakamura, S. ; Toru, T. J. Am. Chem. Soc. 2007, 129, 6394.
(e) Prakash, G. K. S. ; Gurung, L. ; Jog, P. V. ; Tanaka, S. ; Thomas, T. E. ; Ganesh, N. ; Haiges, R. ; Mathew, T. ; Olah, G. A. Chem. Eur. J. 2013, 19, 3579. -
[8]
(a) Furukawa, T. ; Shibata, N. ; Mizuta, S. ; Nakamura, S. ; Toru, T. ; Shiro, M. Angew. Chem., Int. Ed. 2008, 47, 8051.
(b) Zhang, S. ; Zhang, Y. ; Ji, Y. ; Li, H. ; Wang, W. Chem. Commun. 2009, 4886.
(c) Alba, A. -N. ; Companyó, X. ; Moyano, A. ; Rios, R. Chem. Eur. J. 2009, 15, 7035.
(d) Moon, H. W. ; Cho, M. J. ; Kim, D. Y. Tetrahedron Lett. 2009, 50, 4896.
(e) Ullah, F. ; Zhao, G. -L. ; Deiana, L. ; Zhu, M. ; Dziedzic, P. ; Ibrahem, I. ; Hammar, P. ; Sun, J. ; Córdova, A. Chem. Eur. J. 2009, 15, 10013. -
[9]
Ni, C.; Zhang, L.; Hu, J. J. Org. Chem. 2008, 73, 5699. doi: 10.1021/jo702479z
-
[10]
hen, X.; Zhang, W.; Zhang, L.; Luo, T.; Wan, X.; Gu, Y.; Hu, J. Angew. Chem., Int. Ed. 2012, 51, 6966. doi: 10.1002/anie.201202451
-
[11]
Ogasawara, M.; Murakami, H.; Furukawa, T.; Takahashi, T.; Shibata, N. Chem. Commun. 2009, 7366.
-
[12]
(a) Baszczyňski, O. ; Zaneba, Z. Med. Res. Rev. 2013, 33, 1304.
(b) Jindřich, J. ; Hol, A. ; Dvořáková, H. Collect. Czech. Chem. Commun. 1993, 58, 1645.
(c) Kiesewetter, D. O. ; Knudson, K. ; Collins, M. ; Srinivasula, S. ; Lim, E. ; Mascio, M. D. J. Labelled Compd. Radiopharm. 2008, 51, 187. -
[13]
(a) Liang, L. ; Xie, M. -S. ; Wang, H. -X. ; Niu, H. -Y. ; Qu, G. -R. ; Guo, H. -M. J. Org. Chem. 2017, 82, 5966.
(b) Liang, L. ; Xie, M. -S. ; Qin, T. ; Zhu, M. ; Qu, G. -R. ; Guo, H. -M. Org. Lett. 2017, 19, 5212.
(c) Sun, H. -L. ; Chen, F. ; Xie, M. -S. ; Guo, H. -M. ; Qu, G. -R. ; He, Y. -M. ; Fan, Q. -H. Org. Lett. 2016, 18, 2260.
(d) Zhang, D. -J. ; Xie, M. -S. ; Qu, G. -R. ; Gao, Y. -W. ; Guo, H. -M. Org. Lett. 2016, 18, 820.
(e) Xie, M. -S. ; Wang, Y. ; Li, J. -P. ; Du, C. ; Zhang, Y. -Y. ; Hao, E. -J. ; Zhang, Y. -M. ; Qu, G. -R. ; Guo, H. -M. Chem. Commun. 2015, 51, 12451.
(f) Niu, H. -Y. ; Du, C. ; Xie, M. -S. ; Wang, Y. ; Zhang, Q. ; Qu, G. -R. ; Guo, H. -M. Chem. Commun. 2015, 51, 3328.
(g) Wei, T. ; Xie, M. -S. ; Qu, G. -R. ; Niu, H. -Y. ; Guo, H. -M. Org. Lett. 2014, 16, 900. -
[14]
For reviews about synthesis of acyclic nucleoside analogues, see: (a) Xie, M. -S. ; Niu, H. -Y. ; Qu, G. -R. ; Guo, H. -M. Tetrahedron Lett. 2014, 55, 7156.
(b) Guo, H. -M. ; Wu, S. ; Niu, H. -Y. ; Song, G. ; Qu, G. -R. In Chemical Synthesis of Nucleoside Analogues, Ed. : Merino, P., John Wiley & Sons, Hoboken, New Jersey, 2013, p. 103.
-
[1]
-
Table 1. Optimization of the monofluoromethylation of 6-methoxy-purin-9-yl allene (1a) with FBSMa

Entry Catalyst (mol%) Solvent Base (equiv.) Temp. Yieldb/% 2a 3a 1 AgF (10) DMF NaH (2) r.t. 22 14 2 AgF (10) CH2Cl2 NaH (2) r.t. N.R. N.R. 3 AgF (10) CH3CN NaH (2) r.t. 31 26 4 AgF (10) THF NaH (2) r.t. 53 43 5 AgF (10) THF Na2CO3 (2) r.t. 13 16 6 AgF (10) THF NaOtBu (2) r.t. Trace 41 7 AgF (10) THF iPr2EtN (2) r.t. 54 21 8 AgF (10) THF Et3N (2) r.t. 64 28 9 AgF (10) THF Et3N (1.5) r.t. 71 24 10 AgF (10) THF Et3N (1) r.t. 38 11 11 Ag2CO3 (10) THF Et3N (1.5) r.t. 34 11 12 AgSbF6 (10) THF Et3N (1.5) r.t. 42 17 13 Pd(dba)2 (10) THF Et3N (1.5) r.t. N.R. N.R. 14 Pd(OAc)2 (10) THF Et3N (1.5) r.t. N.R. N.R. 15 Pd(PPh3)4 (10) THF Et3N (1.5) r.t. N.R. N.R. 16 AgF (3) THF Et3N (1.5) r.t. 73 23 17 AgF (1) THF Et3N (1.5) r.t. 65 16 18 AgF (3) THF Et3N (1.5) 0 ℃ 59 0 19c AgF (3) THF Et3N (1.5) 0℃ 82 0 a Unless otherwise noted, the reaction conditions were: 1a (0.1 mmol), FBSM (1.1 equiv.), catalyst, solvent (1.0 mL), base for 12 h. b Isolated yield based on 1a. c Reaction time: 48 h, N.R.: no reaction. Table 2. Substrate scope of purin-9-yl allenesa, b



a Reaction conditions: 1a~1j(0.2 mmol), FBSM (1.1 equiv.), AgF (3 mol%), Et3N (1.5 equiv.), THF (2.0 mL) at 0℃ for 48 h.bIsolated yield. -

 扫一扫看文章
扫一扫看文章
计量
- PDF下载量: 10
- 文章访问数: 2856
- HTML全文浏览量: 155


 下载:
下载:



 下载:
下载:

