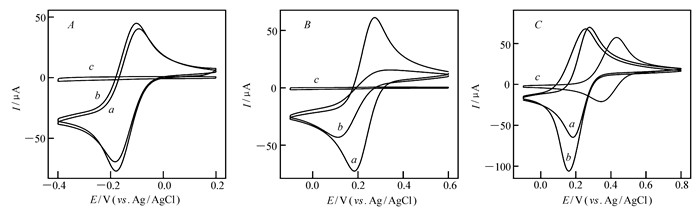
 图1
Ru(NH3)63+(A)、Fe(CN)63-(B)和FcMeOH(C)在ITO(a)、SIM/ITO(b)和CTAB@SIM/ITO(c)电极上的循环伏安曲线
Figure1.
CVs for 0.5 mmol/L Ru(NH3)6Cl3(A), K3[Fe(CN)6](B) and FcMeOH(C) in 0.05 mol/L KHP solution at bare ITO(a), SIM/ITO(b) and CTAB@SIM/ITO(c)
图1
Ru(NH3)63+(A)、Fe(CN)63-(B)和FcMeOH(C)在ITO(a)、SIM/ITO(b)和CTAB@SIM/ITO(c)电极上的循环伏安曲线
Figure1.
CVs for 0.5 mmol/L Ru(NH3)6Cl3(A), K3[Fe(CN)6](B) and FcMeOH(C) in 0.05 mol/L KHP solution at bare ITO(a), SIM/ITO(b) and CTAB@SIM/ITO(c)

Citation: DING Hao, YAN Fei, ZHENG Wenjing, GUO Weiliang, SU Bin. Electrochemical Detection of Antioxidants in Cosmetic Samples Using Mesoporous Silica Film and Confined Micelles[J]. Chinese Journal of Applied Chemistry, 2017, 34(11): 1307-1313. doi: 10.11944/j.issn.1000-0518.2017.11.170251

二氧化硅纳米均孔膜修饰电极检测化妆水中的抗氧化剂
English
Electrochemical Detection of Antioxidants in Cosmetic Samples Using Mesoporous Silica Film and Confined Micelles
-
Key words:
- electrochemical sensor
- / mesoporous silica film
- / antioxidants
- / micelles
- / cosmetic
-
丁基羟基苯甲醚(butylated hydroxyanisole, BHA)和叔丁基对苯二酚(tert-butylhydroquinone, TBHQ)等人工合成的酚类抗氧化剂作为添加剂,已经被广泛地用于延缓或抑制食物、油脂、染料和聚合物等的氧化[1]。此外,它们还用作化妆品中的芳香剂,可以中断脂类的自由基链式反应,达到修复皮肤,提高细胞活力,预防皮肤癌的效果[2]。但是,抗氧化剂的过度使用会威胁人体健康,研究结果表明,BHA的滥用会导致肝损伤[3]。而且,相关法规规定化妆品中的抗氧化剂总量必须控制在10~200 mg/L范围内[4]。因此,发展灵敏的定量检测方法对于控制抗氧化剂的合理使用具有十分重要的意义。
检测合成酚类抗氧化剂的方法有多种,包括分光光度法[3-5]、气相色谱法[6]、高效液相色谱法[7-8]、胶束电动毛细管色谱[2]和气/液相色谱与质谱联用法[9-10]等。这些方法中有些需要昂贵的设备,有些需要费时费力的样品前处理[11]。相比之下,电化学法具有灵敏度高、选择性高、费用低且响应快速等优势[12],是一种较为理想的分析方法。近年来,基于修饰电极的电化学方法受到了广泛关注,各种新材料包括纳米颗粒[13-15]、碳纳米管[16-17]、氧化石墨烯[18]及其复合物[19-21]等,均已被深入研究并成功用于提高电化学传感器的灵敏度和选择性。
二氧化硅纳米均孔膜(silica isoporous membrane, SIM)由周期性排布的均一纳米孔道及其周围的无定形二氧化硅组成。SIM比表面积大,结构高度有序,具有优良的吸附能力和易于修饰的特性[22],使其在电分析传感器领域受到了众多分析化学研究者的关注[23-24]。然而,二氧化硅表面具有亲水的硅羟基,不利于其对溶液相中疏水待测物的富集与检测。为解决这一问题,本课题组最近报道了一种由二氧化硅纳米通道与模板分子十六烷基三甲基溴化铵(cetyltrimethylammonium bromide,CTAB)所构成的二元异质二氧化硅均孔薄膜修饰的氧化铟锡(ITO)电极(简写为CTAB@SIM/ITO)[25-28]。由于纳米通道中疏水胶束的萃取和富集作用,该修饰电极实现了溶液中疏水分子的选择性检测。此外,由于纳米通道超小的尺寸(直径2.3 nm),二氧化硅均孔膜表现出明显的尺寸排阻效应和抗污染性能,实现了未经处理的复杂介质(土壤浊液、牛奶、蜂蜜、果汁、人血清和全血等)中目标分析物的直接检测。本文使用CTAB@SIM/ITO电极,直接检测了化妆品样品中的抗氧化剂BHA和TBHQ。
1 实验部分
1.1 仪器和试剂
CHI 660D型电化学工作站(上海辰华仪器公司); SU8010型场发射扫描电子显微镜(日本日立公司); HT7700型电子显微镜(日本日立公司)。所有溶液均用超纯水(>18.2 MΩ·cm)配制。浓氨水(质量分数25%)、铁氰化钾(K3[Fe(CN)6])、邻苯二甲酸氢钾(Potassium biphthalate, KHP)、丁基羟基苯甲醚(BHA,>99%)和叔丁基对苯二酚(TBHQ,>99%)购买于Aladdin试剂公司。甲醇二茂铁(FcMeOH, 97%)和CTAB(≥ 98%)购买于Alfa Aesar试剂公司。三氯化六氨合钌(Ru(NH3)6Cl3, 98%)和四乙氧基硅氧烷(TEOS,≥ 99.0%)从Sigma试剂公司购买。BHA和TBHQ的储备液用乙醇配制并在4 ℃储存,使用时用磷酸缓冲液(phosphate buffer, PB)稀释至所需浓度。化妆水(千纤草黄瓜水)从浙江大学教育超市购买。氧化铟锡导电玻璃(ITO,方阻<17 ohm/sq,ITO厚度(100±20) nm)使用前依次用1 mol/L NaOH/乙醇溶液,丙酮,乙醇和超纯水超声清洗,每次20 min。
1.2 实验方法
CTAB@SIM/ITO电极依据赵东元课题组报道的Stöber溶液生长法[29]制备得到。制备步骤为:在70 mL水和30 mL乙醇的混合溶液中溶解0.16 g CTAB,搅拌下加入10 μL浓氨水和80 μL TEOS。将ITO导电玻璃浸入上述溶液中60 ℃水浴静置,24 h后取出,大量水冲洗,100 ℃老化过夜,得到CTAB@SIM/ITO。搅拌下将CTAB@SIM/ITO浸入0.1 mol/L盐酸/乙醇溶液中,去除SIM孔道中的CTAB,得到二氧化硅纳米通道修饰的ITO电极(简写为SIM/ITO)。
2 结果与讨论
2.1 CTAB@SIM/ITO电极表征
CTAB@SIM膜的表面形貌和厚度分别用透射电子显微镜和扫描电子显微镜表征(见辅助材料图S1A),SIM表面完整无裂痕,均匀地分布有直径2.3 nm的介孔。CTAB@SIM/ITO的3层结构(见辅助材料图S1B),从上到下分别为CTAB@SIM,ITO和玻璃基底。CTAB@SIM膜的厚度(即胶束的厚度)为92 nm。根据接触角测试的结果(见辅助材料图S2)可知,CTAB胶束疏水碳氢长链的存在使得电极表面更加疏水。
循环伏安(CV)图表明了CTAB@SIM膜的渗透性和胶束的萃取能力。图 1比较了3种氧化还原探针,带正电荷的Ru(NH3)63+(图 1A),带负电荷的Fe(CN)63-(图 1B)以及中性分子FcMeOH(图 1C)的CV曲线。在CTAB@SIM/ITO电极(图 1曲线c)上,只有中性分子FcMeOH得到了一对氧化还原峰,Ru(NH3)63+与Fe(CN)63-均无信号。除去CTAB,即在SIM/ITO电极(图 1曲线b)上,3种氧化还原探针均能在电极上发生氧化还原。CV结果表明,CTAB胶束能够选择性阻碍带电物种进入纳米通道,而中性或疏水性的分子能够顺利通过纳米通道,到达ITO电极表面,进而发生氧化还原反应。Fe(CN)63-在SIM/ITO电极上的电流较裸ITO上的小,说明其氧化还原受到抑制,这是因为带负电的分子和纳米通道内脱质子的硅羟基(等电点2~3[30])相互排斥。以上CV结果与文献[31]报道的一致。
 图1
Ru(NH3)63+(A)、Fe(CN)63-(B)和FcMeOH(C)在ITO(a)、SIM/ITO(b)和CTAB@SIM/ITO(c)电极上的循环伏安曲线
Figure1.
CVs for 0.5 mmol/L Ru(NH3)6Cl3(A), K3[Fe(CN)6](B) and FcMeOH(C) in 0.05 mol/L KHP solution at bare ITO(a), SIM/ITO(b) and CTAB@SIM/ITO(c)
图1
Ru(NH3)63+(A)、Fe(CN)63-(B)和FcMeOH(C)在ITO(a)、SIM/ITO(b)和CTAB@SIM/ITO(c)电极上的循环伏安曲线
Figure1.
CVs for 0.5 mmol/L Ru(NH3)6Cl3(A), K3[Fe(CN)6](B) and FcMeOH(C) in 0.05 mol/L KHP solution at bare ITO(a), SIM/ITO(b) and CTAB@SIM/ITO(c)
2.2 BHA和TBHQ在CTAB@SIM/ITO电极上的电化学行为
图 2分别为10 mg/L BHA和TBHQ在0.1 mol/L PB(pH=7.0)中的CV(图 2A、图 2C)和DPV(图 2B、图 2D)曲线。BHA在CTAB@SIM/ITO电极上1.08 V处的氧化峰电流值明显高于其他两种电极;而-0.355 V处的还原峰仅在CTAB@SIM/ITO电极上较明显。上述结果说明,CTAB胶束能够通过疏水作用,有效地从水相溶液中萃取疏水小分子BHA;相比其它两种电极,CTAB@SIM/ITO对BHA的电流响应更灵敏。类似的结果也在TBHQ的检测中得到(图 2B、2D)。人们普遍认为BHA和TBHQ的电化学氧化是一个两电子转移过程,伴随着对应的醌的生成[1]。在不同扫速下(50~300 mV/s)对含有10 mg/L BHA的溶液进行循环伏安测试,并将所得氧化峰电流对扫速的平方根作图,得到了良好的线性回归曲线(见辅助材料图S3),证明BHA在电极表面的电化学过程受扩散控制,同时表明待测物分子能够很好地“溶解”在CTAB胶束内,并自由扩散到ITO电极表面。
2.3 BHA和TBHQ的电化学检测
采用机械搅拌方式作为预富集手段(见辅助材料图S4),搅拌速度为400 r/min,搅拌时间为30 s时,能够得到较高的氧化峰电流。在优化的条件下,分别记录CTAB@SIM/ITO电极对BHA和TBHQ的DPV响应并用于定量分析。BHA的氧化峰电流随浓度增加明显升高(图 3A),对应的标准曲线为图 3B,低、高浓度段线性范围分别为0.10~3.07 mg/L和3.07~15.0 mg/L,灵敏度分别为(0.891±0.013) μA·L/mg(R=0.9986)(低浓度)和(0.443±0.015) μA·L/mg(R=0.9965)(高浓度)。检测限为(3.11±0.05)μg/L。TBHQ的电化学检测在相同的条件下完成,结果(包括灵敏度、线性范围、检测限和R)列于表 1。图 3C和3D为TBHQ的DPV曲线和标准曲线。我们尝试了BHA和TBHQ的同时检测,但两种物质的峰电位差小于200 mV,表明两组峰间有重叠,阻碍了混合体系中单一组分的正确定量。
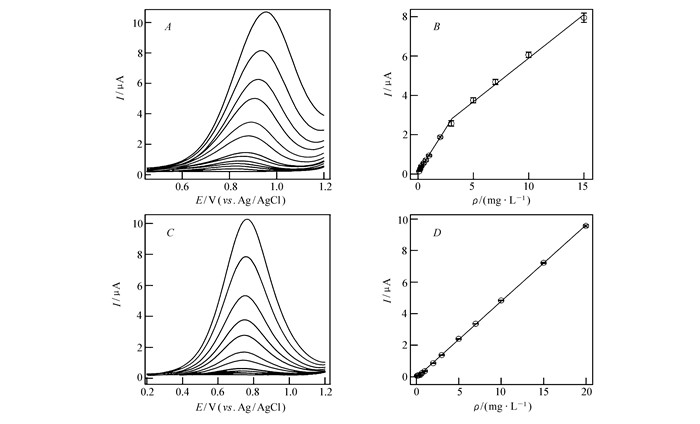 图3
CTAB@SIM/ITO电极检测0.1 mol/L PB中的不同浓度BHA(A, B)和TBHQ(C, D)DPV曲线(A, B)及相应峰电流-浓度关系曲线(B, D)
Figure3.
DPVs for various concentrations of BHA(A) and TBHQ(C) at the CTAB@SIM/ITO electrode in 0.1 mol/L PB solution(pH=7.0); Plot of the anodic peak current as a function of BHA(B) and TBHQ(D) concentration obtained at the CTAB@SIM/ITO electrode. Each experiment was done in triplicate
图3
CTAB@SIM/ITO电极检测0.1 mol/L PB中的不同浓度BHA(A, B)和TBHQ(C, D)DPV曲线(A, B)及相应峰电流-浓度关系曲线(B, D)
Figure3.
DPVs for various concentrations of BHA(A) and TBHQ(C) at the CTAB@SIM/ITO electrode in 0.1 mol/L PB solution(pH=7.0); Plot of the anodic peak current as a function of BHA(B) and TBHQ(D) concentration obtained at the CTAB@SIM/ITO electrode. Each experiment was done in triplicate
Analytes Sensitivity/(μA·L·mg-1) Dynamic range/(mg·L-1) Limit of detection/(μg·L-1) R BHA 0.891±0.013 0.10~3.07 3.11±0.05 0.998 6 0.443±0.015 3.07~15.0 0.996 5 TBHQ 0.481±0.002 0.10~20.0 61.0±0.3 0.999 8 a.Average of three determinations. 2.4 BHA和TBHQ在实际样品中的检测
限定在纳米通道内的胶束提供了疏水的微环境,纳米通道超小的尺寸使其具有尺寸筛分能力,这二者均抑制了复杂样品中的大分子或溶液中的非目标分子对电极表面的污染。因此,CTAB@SIM/ITO电极的抗污染能力是可以预见的。
图 4比较了CTAB@SIM/ITO和裸ITO电极分别在PB溶液和稀释10倍的化妆水样品中检测BHA的标准曲线。在PB溶液中,裸ITO电极上得到了BHA浓度和峰电流的标准曲线,两段线性分别为0.50~3.00 mg/L和3.00~10.0 mg/L,线性范围较CTAB@SIM/ITO电极窄,且灵敏度(0.029±0.0126) μA·L/mg(低浓度)和(0.016±0.005) μA·L/mg(高浓度)远低于CTAB@SIM/ITO电极。在化妆水样品中,ITO电极上不能得到峰电流与BHA浓度的对应标准曲线,而在CTAB@SIM/ITO电极上,我们得到了与缓冲溶液中几乎相同的灵敏度(图 4曲线a和b)。此外,使用标准加入法,得到了BHA和TBHQ在化妆水样品中的回收率(表 2),分别为96.8%和104%,RSD在2.2%内,表明了该方法用于实际样品检测的可行性。
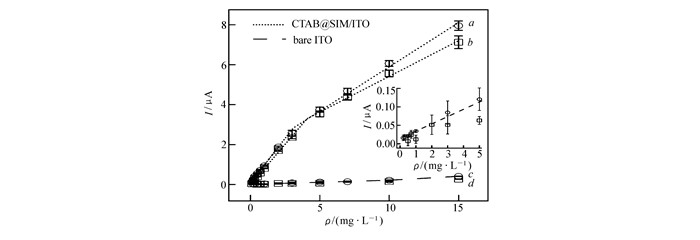 图4
CTAB@SIM/ITO电极和裸ITO电极检测BHA和TBHQ的标准曲线对比
Figure4.
Calibration curves for BHA(0.1~15 mg/L) in 0.1 mol/L PB solution(pH=7.0)(a, c) and in cosmetic sample(b, d) at modified(a, b) and bare(c, d) ITO electrodes. Each experiment was done in triplicate. Inset:expended view of the bare ITO in 0.1 mol/L PB(pH=7.0)(○) and cosmetic samples(□) in the low concentration region
图4
CTAB@SIM/ITO电极和裸ITO电极检测BHA和TBHQ的标准曲线对比
Figure4.
Calibration curves for BHA(0.1~15 mg/L) in 0.1 mol/L PB solution(pH=7.0)(a, c) and in cosmetic sample(b, d) at modified(a, b) and bare(c, d) ITO electrodes. Each experiment was done in triplicate. Inset:expended view of the bare ITO in 0.1 mol/L PB(pH=7.0)(○) and cosmetic samples(□) in the low concentration region
Analytes Added/(mg·L-1) Found/(mg·L-1) Recovery/% RSD/% BHA 1.50 1.56 104 2.2 TBHQ 12.5 12.1 96.8 1.0 a.Average of three determinations. 3 结论
本文基于二氧化硅纳米均孔膜修饰的ITO电极,提出了一种高灵敏度的电化学方法,在化妆水样品中定量检测了两种合成酚类抗氧化剂丁基羟基苯甲醚(BHA)和叔丁基对苯二酚(TBHQ)。均孔膜上的纳米通道作为选择渗透性的屏障,排除了较大尺寸物质的干扰;孔道内的十六烷基三甲基溴化铵胶束萃取水溶液中疏水性的抗氧化剂分子。该电极在磷酸盐缓冲液和化妆品样品中进行分析测试的可靠性和实际应用能力均得到验证。此外,该方法费用低、更省时,具有应用于常规质量监测的潜力。
辅助材料(Supporting Information)[CTAB@SIM膜的TEM和SEM表征;CTAB@SIM/ITO电极的疏水性及电化学性能]可以免费从本刊网站(http://yyhx.ciac.jl.cn/)下载。
-
-
[1]
Ziyatdinova G, Os'kina K, Ziganshina E. Simultaneous Determination of TBHQ and BHA on a MWNT-Brij® 35 Modified Electrode in Micellar Media[J]. Anal Methods, 2015, 7(19): 8344-8351. doi: 10.1039/C5AY01973G
-
[2]
Guan Y, Chu Q, Fu L. Determination of Antioxidants in Cosmetics by Micellar Electrokinetic Capillary Chromatography with Electrochemical Detection[J]. J Chromatogr A, 2005, 1074(1/2): 201-204.
-
[3]
Wang J Y, Wu H L, Chen Y. Quantitative Determination of Butylated Hydroxyanisole and n-Propyl Gallate in Cosmetics Using Three-Dimensional Fluorescence Coupled with Second-Order Calibration[J]. Talanta, 2013, 116: 347-353. doi: 10.1016/j.talanta.2013.05.028
-
[4]
Capitan-Vallvey L F, Valencia M C, Nicolas E A. Monoparameter Sensors for the Determination of the Antioxidants Butylated Hydroxyanisole and n-Propyl Gallate in Foods and Cosmetics by Flow Injection Spectrophotometry[J]. Analyst, 2001, 126(6): 897-902. doi: 10.1039/b101162f
-
[5]
Capitan-Vallvey L F, Valencia M C, Nicolas E A. Simple Resolution of Butylated Hydroxyanisole and n-Propyl Gallate in Fatty Foods and Cosmetics Samples by Flow-Injection Solid-Phase Spectrophotometry[J]. J Food Sci, 2003, 68(5): 1595-1599. doi: 10.1111/jfds.2003.68.issue-5
-
[6]
Cacho J I, Campillo N, Viñas P. Determination of Synthetic Phenolic Antioxidants in Edible Oils Using Microvial Insert Large Volume Injection Gas-Chromatography[J]. Food Chem, 2016, 200: 249-254. doi: 10.1016/j.foodchem.2016.01.026
-
[7]
Chen M, Hu X J, Tai Z G. Determination of Four Synthetic Phenolic Antioxidants in Edible Oils by High-Performance Liquid Chromatography with Cloud Point Extraction Using Tergitol TMN-6[J]. Food Anal Methods, 2013, 6(1): 28-35. doi: 10.1007/s12161-012-9413-7
-
[8]
Biparva P, Ehsani M, Hadjmohammadi M R. Dispersive Liquid-Liquid Microextraction Using Extraction Solvents Lighter than Water Combined with High Performance Liquid Chromatography for Determination of Synthetic Antioxidants in Fruit Juice Samples[J]. J Food Compos Anal, 2012, 27(1): 87-94. doi: 10.1016/j.jfca.2012.04.002
-
[9]
Touati R, Santos S A O, Rocha S M. The Potential of Cork from Quercus Suber L. Grown in Algeria as a Source of Bioactive Lipophilic and Phenolic Compounds[J]. Ind Crop Prod, 2015, 76: 936-945. doi: 10.1016/j.indcrop.2015.07.074
-
[10]
Rodil R, Quintana J B, Basaglia G. Determination of Synthetic Phenolic Antioxidants and Their Metabolites in Water Samples by Downscaled Solid-Phase Extraction, Silylation and gas Chromatography-Mass Spectrometry[J]. J Chromatogr A, 2010, 1217(41): 6428-6435. doi: 10.1016/j.chroma.2010.08.020
-
[11]
Wang P, Han C, Zhou F. Electrochemical Determination of Tert-Butylhydroquinone and Butylated Hydroxyanisole at Choline Functionalized Film Supported Graphene Interface[J]. Sens Actuators B, 2016, 224: 885-891. doi: 10.1016/j.snb.2015.10.098
-
[12]
Sousa Carvalho R M, Yotsumoto Neto S, Carvalho Silva F. A Sensitive Sensor Based on CuTSPc and Reduced Graphene Oxide for Simultaneous Determination of the BHA and TBHQ Antioxidants in Biodiesel Samples[J]. Electroanalysis, 2016, 28(12): 2930-2938. doi: 10.1002/elan.201600187
-
[13]
Lin X, Ni Y, Kokot S. Glassy Carbon Electrodes Modified with Gold Nanoparticles for the Simultaneous Determination of Three Food Antioxidants[J]. Anal Chim Acta, 2013, 765: 54-62. doi: 10.1016/j.aca.2012.12.036
-
[14]
Niu X, Yang W, Guo H. A Novel Strategy for the Detection of tert-Butylhydroquinone Based on Graphene Quantum Dots and Silver Nanoparticle Modified Glass Carbon Electrode[J]. Can J Chem, 2015, 93(6): 648-654. doi: 10.1139/cjc-2014-0339
-
[15]
Roushani M, Sarabaegi M. Electrochemical Detection of Butylated Hydroxyanisole Based on Glassy Carbon Electrode Modified by Iridium Oxide Nanoparticles[J]. J Electroanal Chem, 2014, 717: 147-152.
-
[16]
Caramit R P, Antunes Araújo A S, Fogliatto D K. Carbon-Nanotube-Modified Screen-Printed Electrodes, a Cationic Surfactant, and a Peak Deconvolution Procedure:Alternatives to Provide Satisfactory Simultaneous Determination of Three Synthetic Antioxidants in Complex Samples[J]. Anal Methods, 2015, 7(9): 3764-3771. doi: 10.1039/C4AY02875A
-
[17]
Caramit R P, de Freitas Andrade A G, Gomes de Souza J B. A New Voltammetric Method for the Simultaneous Determination of the Antioxidants TBHQ and BHA in Biodiesel Using Multi-Walled Carbon Nanotube Screen-Printed Electrodes[J]. Fuel, 2013, 105: 306-313. doi: 10.1016/j.fuel.2012.06.062
-
[18]
Gan T, Zhao A X, Wang S H. Hierarchical Triple-Shelled Porous Hollow Zinc Oxide Spheres Wrapped in Graphene Oxide as Efficient Sensor Material for Simultaneous Electrochemical Determination of Synthetic Antioxidants in Vegetable Oil[J]. Sens Actuators B, 2016, 235: 707-716. doi: 10.1016/j.snb.2016.05.137
-
[19]
Cui M, Liu S, Lian W J. A Molecularly-Imprinted Electrochemical Sensor Based on a Graphene-Prussian Blue Composite-Modified Glassy Carbon Electrode for the Detection of Butylated Hydroxyanisole in Foodstuffs[J]. Analyst, 2013, 138(20): 5949-5955. doi: 10.1039/c3an01190a
-
[20]
dos Santos Moretti E, de Oliveira F M, Scheel G L. Synthesis of Surface Molecularly Imprinted Poly(methacrylic acid-hemin) on Carbon Nanotubes for the Voltammetric Simultaneous Determination of Antioxidants from Lipid Matrices and Biodiesel[J]. Electrochim Acta, 2016, 212: 322-332. doi: 10.1016/j.electacta.2016.06.174
-
[21]
Yue X, Song W, Zhu W. In Situ Surface Electrochemical Co-Reduction Route Towards Controllable Construction of AuNPs/ERGO Electrochemical Sensing Platform for Simultaneous Determination of BHA and TBHQ[J]. Electrochim Acta, 2015, 182: 847-855. doi: 10.1016/j.electacta.2015.09.162
-
[22]
Rao H, Wang X, Du X. Mini Review:Electroanalytical Sensors of Mesoporous Silica Materials[J]. Anal Lett, 2013, 46(18): 2789-2812. doi: 10.1080/00032719.2013.816962
-
[23]
Serrano M B, Despas C, Herzog G. Mesoporous Silica Thin Films for Molecular Sieving and Electrode Surface Protection Against Biofouling[J]. Electrochem Commun, 2015, 52: 34-36. doi: 10.1016/j.elecom.2015.01.010
-
[24]
Walcarius A, Mercier L. Mesoporous Organosilica Adsorbents:Nanoengineered Materials for Removal of Organic and Inorganic Pollutants[J]. J Mater Chem, 2010, 20(22): 4478. doi: 10.1039/b924316j
-
[25]
Sun Q, Yan F, Yao L. Anti-Biofouling Isoporous Silica-Micelle Membrane Enabling Drug Detection in Human Whole Blood[J]. Anal Chem, 2016, 88(17): 8364-8368. doi: 10.1021/acs.analchem.6b02091
-
[26]
Yao L, Yan F, Su B. Highly Ordered Surfactant Micelles Function as the Extraction Matrix for Direct Electrochemical Detection of Halonitrobenzenes at the ppb Level[J]. Analyst, 2016, 141(7): 2303-2307. doi: 10.1039/C5AN02439K
-
[27]
Yan F, Su B. Tailoring Molecular Permeability of Nanochannel-Micelle Membranes for Electrochemical Analysis of Antioxidants in Fruit Juices without Sample Treatment[J]. Anal Chem, 2016, 88(22): 11001-11006. doi: 10.1021/acs.analchem.6b02823
-
[28]
Zheng W, Yan F, Su B. Electrochemical Determination of Chloramphenicol in Milk and Honey Using Vertically Ordered Silica Mesochannels and Surfactant Micelles as the Extraction and Anti-Fouling Element[J]. J Electroanal Chem, 2016, 781: 383-388. doi: 10.1016/j.jelechem.2016.04.017
-
[29]
Teng Z, Zheng G, Dou Y. Highly Ordered Mesoporous Silica Films with Perpendicular Mesochannels by a Simple Stober-Solution Growth Approach[J]. Angew Chem Int Ed, 2012, 51(9): 2173-2177. doi: 10.1002/anie.201108748
-
[30]
Etienne M, Quach A, Grosso D. Molecular Transport into Mesostructured Silica Thin Films:Electrochemical Monitoring and Comparison Between p6m, P63/mmc, and Pm3n Structures[J]. Chem Mater, 2007, 19(4): 844-856. doi: 10.1021/cm0625068
-
[31]
Yan F, Lin X, Su B. Vertically Ordered Silica Mesochannel Films:Electrochemistry and Analytical Applications[J]. Analyst, 2016, 141(12): 3482-3495. doi: 10.1039/C6AN00146G
-
[1]
-
图 3 CTAB@SIM/ITO电极检测0.1 mol/L PB中的不同浓度BHA(A, B)和TBHQ(C, D)DPV曲线(A, B)及相应峰电流-浓度关系曲线(B, D)
Figure 3 DPVs for various concentrations of BHA(A) and TBHQ(C) at the CTAB@SIM/ITO electrode in 0.1 mol/L PB solution(pH=7.0); Plot of the anodic peak current as a function of BHA(B) and TBHQ(D) concentration obtained at the CTAB@SIM/ITO electrode. Each experiment was done in triplicate
图 4 CTAB@SIM/ITO电极和裸ITO电极检测BHA和TBHQ的标准曲线对比
Figure 4 Calibration curves for BHA(0.1~15 mg/L) in 0.1 mol/L PB solution(pH=7.0)(a, c) and in cosmetic sample(b, d) at modified(a, b) and bare(c, d) ITO electrodes. Each experiment was done in triplicate. Inset:expended view of the bare ITO in 0.1 mol/L PB(pH=7.0)(○) and cosmetic samples(□) in the low concentration region
表 1 BHA和TBHQ的检测结果
Table 1. Detection results of BHA and TBHQ in buffer solutiona
Analytes Sensitivity/(μA·L·mg-1) Dynamic range/(mg·L-1) Limit of detection/(μg·L-1) R BHA 0.891±0.013 0.10~3.07 3.11±0.05 0.998 6 0.443±0.015 3.07~15.0 0.996 5 TBHQ 0.481±0.002 0.10~20.0 61.0±0.3 0.999 8 a.Average of three determinations. 表 2 BHA和TBHQ在实际样品中的回收率(n=3)
Table 2. Recovery of BHA and TBHQ in real samplesa
Analytes Added/(mg·L-1) Found/(mg·L-1) Recovery/% RSD/% BHA 1.50 1.56 104 2.2 TBHQ 12.5 12.1 96.8 1.0 a.Average of three determinations. -

 扫一扫看文章
扫一扫看文章
计量
- PDF下载量: 8
- 文章访问数: 1191
- HTML全文浏览量: 239


 下载:
下载:



 下载:
下载:

