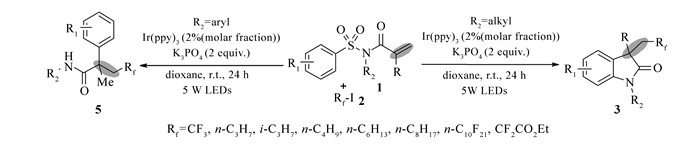
 Scheme1.
Synthesis of perfluorinated oxindoles or α-aryl-β-perfluorobutyl amides
Scheme1.
Synthesis of perfluorinated oxindoles or α-aryl-β-perfluorobutyl amides

Citation: LI Zengzeng, YUAN Li, WANG Liangneng, WANG Shuowen, YU Jian, XIA Lyulu, DENG Youlin, TANG Shi. Visible Light Induced Desulfonylative Annulation Cascade of N-Sulfonylmethacrylamides[J]. Chinese Journal of Applied Chemistry, 2017, 34(11): 1287-1294. doi: 10.11944/j.issn.1000-0518.2017.11.170054

可见光诱导的共轭磺酰胺串联脱砜/环化反应
English
Visible Light Induced Desulfonylative Annulation Cascade of N-Sulfonylmethacrylamides
-
Key words:
- N-sulfonylmethacrylamides
- / perfluoroalkylation
- / cascade cyclization
- / photoredox catalysis
- / oxindoles
-
含氟有机分子广泛存在于很多药物和有机农药化学品中[1]。在复杂的分子骨架中引入含氟基团已经被广泛地用于医药化学中[2]。值得注意的是,利用各种氟试剂(例如, Umemoto试剂、Togni试剂和CF3SO2Cl等)进行烯烃双官能团化,一步引入两个新的官能基团,作为有效的有机氟化合物制备途径在过去的几年中得到许多有机化学家的关注[3-5]。2013年,Nevado课题组[6]报道了芳基磺酰胺三氟甲基化合成α-芳基-β-三氟甲基酰胺。2015年,Xia课题组[7]也报道了一例类似反应,他们利用CF3SO2Cl作为三氟甲基自由基源,在钌光催化剂的催化作用下,实现芳基磺酰胺自由基串联加成/环化反应。但是,这些方法只限于在分子骨架中引入简单的氟代甲基基团(例如二氟甲基或三氟甲基)。因此,在利用苯磺酰胺串联脱磺/环合合成含氟吲哚酮反应中,增加引入的含氟基团的多样性(如引入各种全氟烷基化基团),目前仍然具有重要的合成意义。由于其具有方便、绿色、安全等优点,可见光催化在过去的几年中,已经发展成为一种强大的合成工具被用于现代有机合成中[8]。全氟碘/溴代烷基试剂是有机氟化学常用的试剂,相对于其它常见烷基试剂(例如, RfSO2Cl(Rf为全氟烷基基团)和Togni试剂)来说比较便宜,通过商业渠道也容易得到[9-14]。基于以上研究背景,以及我们对可见光诱导全氟烷基化研究的兴趣[15],我们拟以全氟碘/溴代烷基试剂作为全氟烷基自由源,通过可见光诱导苯磺酰胺脱磺全氟烷基化,在温和的反应条件下合成各种全氟烷基化吲哚酮及α-芳基酰胺类化合物,快速引入CF3、n-C3F7、i-C3F7、n-C4F9、n-C6F13、n-C8F17、n-C10F21和CF2CO2Et(Et:乙基)等一系列含氟基团(Scheme 1)。
1 实验部分
1.1 仪器和试剂
实验中所用的芳基磺酰氯、草酰氯、全氟碘/溴代烷基试剂和面式-三(2-苯基吡啶)合铱(fac-Ir(ppy)3)等均购自阿拉丁试剂公司或百灵威试剂公司,其它试剂(购自安耐吉试剂公司)除标明外均为分析纯。实验中所需的原料芳基磺酰胺1a~1m参照相关文献[6]报道方法合成得到。
AVANCE 400 MHz型超导傅里叶数字化核磁共振仪(瑞士Bruker公司);GC-MS-QP2010型质谱仪(日本岛津公司);XT5A型显微熔点仪(北京市科仪电光学仪器厂);RE-52AA型旋转蒸发仪(上海亚荣生化仪器厂)。
1.2 目标产物3的合成
以目标产物3a的合成为例,取原料N-正丁基-N-甲基丙烯酰基-4-甲基苯磺酰胺1a(88.5 mg, 0.3 mmol)溶于2.0 mL二氧六环中,加入fac-Ir(ppy)3(3.6 mg, 2.0%摩尔分数)和K3PO4(127.2 mg, 0.6 mmol),再加入C4F9I(207.5 mg, 0.6 mmol),并使用N2气保护,在5 W蓝色发光二极管(LED)灯照射下,搅拌反应24 h。经TLC检验反应完全后,经柱层析分离,石油醚-乙酸乙酯(体积比7:1)梯度洗脱,得到目标产物3a, 为一种油状液体,产率为78%。
1.3 产物的表征数据
产物3a:黄色油状物; 1H NMR(400 MHz, CDCl3), δ:7.18~7.13(m, 1H), 6.89(d, J=7.6 Hz, 1H), 6.71(s, 1H), 3.76(dt, J=14.4, 7.2 Hz, 1H), 3.70~3.61(m, 1H), 2.86(dd, J=35.4, 15.1 Hz, 1H), 2.61~2.49(m, 1H), 2.40(s, 3H), 1.69~1.61(m, 2H), 1.43~1.38(m, 5H), 0.96(t, J=7.3 Hz, 3H); 13C NMR(101 MHz, CDCl3), δ:178.7, 142.3, 138.5, 128.5, 123.4, 122.8, 121.3~110.1(m), 109.7, 43.9, 39.7, 36.8(d, J=20.5 Hz), 29.3, 26.2, 21.9, 20.1, 13.7;19F NMR(376 MHz, CDCl3), δ:-81.0(t, J=8.0 Hz, 3F), -108.8(A-B, JF-F=268 Hz, 1F), -114.9(A-B, JF-F=273 Hz, 1F), -124.6(br, 2F), -125.2~ -126.3(m, 2F); HRMS m/z(ESI-TOF)计算值C19H21F9NO [M+H]+ 450.1474, 实测值:450.1476。
产物3b:黄色油状物; 1H NMR(400 MHz, CDCl3), δ:7.15(d, J=7.8 Hz, 1H), 6.88(d, J=5.0 Hz, 1H), 4.62(dt, J=14.0, 7.0 Hz, 1H), 2.87(dd, J=35.3, 16.2 Hz, 1H), 2.62~2.49(m, 1H), 2.39(s, 3H), 1.48(dd, J=7.0, 5.0 Hz, 6H), 1.38(s, 3H); 13C NMR(101 MHz, CDCl3), δ:178.6, 141.6, 138.2, 128.8, 123.5, 122.6, 122.1-111.9(m), 111.1, 43.9, 37.0(t, J=20.3 Hz), 26.3, 22.0, 19.2, 19.0;19F NMR(376 MHz, CDCl3), δ:-81.1(t, J=8.0 Hz, 3F), -108.7(A-B, JF-F=268 Hz, 1F), -114.9(A-B, JF-F=273 Hz, 1F), -124.6(br, 2F), -125.2~ -126.2(m, 2F); HRMS m/z(ESI)计算值C18H19F9NO [M+H]+ 436.1318, 实测值:436.1320。
产物3c:黄色油状物; 1H NMR(400 MHz, CDCl3), δ:7.17(d, J=8.2 Hz, 1H), 6.58(dd, J=8.2, 2.2 Hz, 1H), 6.48(d, J=2.2 Hz, 1H), 3.84(s, 3H), 3.79~3.70(m, 1H), 3.68~3.60(m, 1H), 2.84(dd, J=35.2, 15.2 Hz, 1H), 2.64~2.48(m, 1H), 1.65(tdd, J=10.4, 7.6, 2.9 Hz, 2H), 1.42~1.35(m, 5H), 0.95(t, J=7.3 Hz, 3H); 13C NMR(101 MHz, CDCl3), δ:179.1, 160.2, 143.5, 124.3, 123.4, 120.8~110.2(m), 105.9, 96.9, 55.4, 43.7, 39.9, 36.9(t, J=20.1 Hz), 29.2, 26.2, 20.0, 13.7;19F NMR(376 MHz, CDCl3), δ:-81.1(t, J=9.8 Hz, 3F), -108.8(A-B, JF-F=262.6 Hz, 1F), -113.7(A-B, JF-F=269.3 Hz, 1F), -124.7(br, 2F), -125.1~ -126.8(m, 2F); HRMS m/z(ESI)计算值C19H21F9NO2 [M+H]+ 466.1424, 实测值:466.1426。
产物3d:黄色油状物; 1H NMR(400 MHz, CDCl3), δ:7.40~7.33(m, 2H), 7.09(s, 1H), 3.81(dt, J=14.4, 7.3 Hz, 1H), 3.76~3.65(m, 1H), 2.94(dd, J=35.1, 15.4 Hz, 1H), 2.74~2.51(m, 1H), 1.71~1.61(m, 2H), 1.44(s, 3H), 1.42~1.37(m, 2H), 0.97(t, J=7.3 Hz, 3H); 13C NMR(101 MHz, CDCl3), δ:178.1, 142.9, 135.3, 130.9(q, J=32.6 Hz), 124.3(q, J=272.8 Hz), 122.6, 119.4(q, J=4.1 Hz), 119.3~108.5(m), 105.4(q, J=3.8 Hz), 44.1(d, J=1.9 Hz), 36.8(t, J=20.5 Hz), 29.1, 26.1, 20.1, 13.6;19F NMR(376 MHz, CDCl3), δ:-63.5(s, 3F), -81.2(t, J=9.8 Hz, 3F), -107.3(A-B, JF-F=273.3 Hz, 1F), -112.7(A-B, JF-F=273.0 Hz, 1F), -124.9(br, 2F), -125.1~ -126.7(m, 2F); HRMS m/z(ESI)计算值C19H18F12NO [M+H]+ 504.1192, 实测值:504.1194。
产物3e:黄色油状物; 1H NMR(400 MHz, CDCl3), δ:7.69(dd, J=8.4, 1.1 Hz, 1H), 7.47(d, J=7.4 Hz, 1H), 7.17(dd, J=8.3, 7.5 Hz, 1H), 3.96(ddd, J=15.0, 8.7, 6.5 Hz, 1H), 3.82~3.74(m, 1H), 2.99(dd, J=34.7, 15.4 Hz, 1H), 2.74~2.52(m, 1H), 1.48(s, 3H), 1.42~1.36(m, 2H), 1.33~1.26(m, 2H), 0.90(t, J=7.2 Hz, 3H); 13C NMR(101 MHz, CDCl3), δ:178.9, 136.2, 135.1, 127.3, 124.6, 122.1, 121.2~103.1(m), 43.0, 42.2, 37.2(t, J=20.3 Hz), 29.5, 26.5, 19.7, 13.6;19F NMR(376 MHz, CDCl3), δ:-81.2(t, J=9.9 Hz, 3F), -108.0(A-B, JF-F=273.6 Hz, 1F), -112.8(A-B, JF-F=274.3 Hz, 1F), -124.9(br, 2F), -125.1~ -126.9(m, 2F); HRMS m/z(ESI)计算值C18H18F9N2O3 [M+H]+ 481.1169, 实测值:481.1171。
产物3f:黄色油状物; 1H NMR(400 MHz, CDCl3), δ:7.40~7.33(m, 2H), 7.09(s, 1H), 3.81(dt, J=14.5, 7.3 Hz, 1H), 3.75~3.65(m, 1H), 2.93(dd, J=35.1, 15.4 Hz, 1H), 2.73~2.51(m, 1H), 1.70~1.62(m, 2H), 1.44(s, 3H), 1.43~1.37(m, 2H), 0.97(t, J=7.3 Hz, 3H); 13C NMR(101 MHz, CDCl3), δ:178.1, 142.9, 135.2, 130.9(q, J=32.5 Hz), 124.5(q, J=273.0 Hz), 123.9, 119.4(q, J=4.2 Hz), 119.1~107.3(m), 105.4(q, J=3.7 Hz), 44.0, 40.1, 36.6(t, J=20.9 Hz), 29.1, 26.0, 20.0, 13.7;19F NMR(376 MHz, CDCl3), δ:-63.5(s, 3F), -81.0(t, J=10.0 Hz, 3F), -109.5~-112.4(m, 2F), -127.8~-129.1(m, 2F); HRMS m/z(ESI)计算值C18H18F10NO2 [M+H]+ 454.1224, 实测值:454.1226。
产物3g:黄色固体; mp 86.0~86.7 ℃; 1H NMR (400 MHz, CDCl3), δ:7.34(s, 2H), 7.06(s, 1H), 3.79(dt, J=14.5, 7.4 Hz, 1H), 3.73~3.63(m, 1H), 2.93(t, J=16.3 Hz, 1H), 2.69(t, J=15.2 Hz, 1H), 1.69~1.60(m, 2H), 1.43(s, 3H), 1.39(dd, J=14.5, 7.3 Hz, 2H), 0.98(t, J=7.3 Hz, 3H); 13C NMR(101 MHz, CDCl3), δ:177.4, 142.9, 135.1, 130.9(d, J=32.6 Hz), 123.9, 122.6, 119.2(q, J=4.1 Hz), 118.1-109.2(m), 105.2(q, J=3.9 Hz), 45.4(d, J=4.1 Hz), 40.1, 33.7, 33.5, 29.1, 27.9, 20.1, 13.6;19F NMR(376 MHz, CDCl3), δ:-62.6(s, 3F), -76.46~-78.02(m, 6F), -184.0~-188.0(m, 1F); HRMS m/z(ESI)计算值C18H18F10NO [M+H]+ 454.1224, 实测值:454.1226。
产物3h:黄色固体; mp 45.3~46.1 ℃; 1H NMR(400 MHz, CDCl3), δ:7.15(d, J=7.6 Hz, 1H), 6.89(dd, J=7.6, 0.5 Hz, 1H), 6.71(s, 1H), 3.80~3.71(m, 1H), 3.70~3.60(m, 1H), 2.86(dd, J=35.4, 15.2 Hz, 1H), 2.71~2.50(m, 1H), 2.40(s, 3H), 1.70~1.60(m, 2H), 1.43~1.38(m, 5H), 0.96(t, J=7.3 Hz, 3H); 13C NMR(101 MHz, CDCl3), δ:178.8, 142.3, 138.5, 128.5, 123.4, 122.9, 120.1~110.5(m), 109.7, 43.9, 39.9, 36.8(d, J=20.5 Hz), 29.3, 26.2, 21.9, 20.1, 13.7;19F NMR(376 MHz, CDCl3), δ:-81.0(t, J=8.1 Hz, 3F), -108.6(A-B, JF-F=267 Hz, 1F), -114.8(A-B, JF-F=263 Hz, 1F), -121.9(br, 2F), -122.8(br, 2F), -123.6(br, 2F), -125.2~-126.8(m, 2F); HRMS m/z(ESI)计算值C21H21F13NO [M+H]+ 550.1411, 实测值:550.1413。
产物3i:黄色固体; mp 76.4~77.3 ℃; 1H NMR(400 MHz, CDCl3), δ:7.15(d, J=7.6 Hz, 1H), 6.88(d, J=7.4 Hz, 1H), 6.71(s, 1H), 3.76(dt, J=14.5, 7.1 Hz, 1H), 3.70~3.60(m, 1H), 2.86(dd, J=35.2, 15.2 Hz, 1H), 2.63~2.48(m, 1H), 2.40(s, 3H), 1.65(ddd, J=10.4, 7.7, 2.8 Hz, 2H), 1.44~1.34(m, 5H), 0.96(t, J=7.3 Hz, 3H); 13C NMR(101 MHz, CDCl3), δ:178.8, 142.3, 138.5, 128.5, 123.4, 122.9, 120.7~110.5(m), 109.7, 43.9, 39.9, 36.9(t, J=20.2 Hz), 29.3, 26.2, 21.9, 20.1, 13.7;19F NMR(376 MHz, CDCl3), δ:-80.9(t, J=8.0 Hz, 3F), -108.9(A-B, JF-F=269 Hz, 1F), -114.0(A-B, JF-F=270 Hz, 1F), -121.8(br, 2F), -122.1(br, 4F), -122.9(br, 2F), -123.8(br, 2F), -126.1~-126.8(m, 2F); HRMS m/z(ESI)计算值C23H21F17NO [M+H]+ 650.1347, 实测值:650.1349。
产物3j:黄色固体; mp 71.5~72.5 ℃.1H NMR(400 MHz, CDCl3), δ:7.15(d, J=7.6 Hz, 1H), 6.88(d, J=7.6 Hz, 1H), 6.71(s, 1H), 3.76(dt, J=14.4, 7.2 Hz, 1H), 3.71~3.61(m, 1H), 2.86(dd, J=35.2, 15.2 Hz, 1H), 2.61~2.48(m, 1H), 2.40(s, 3H), 1.68~1.62(m, 2H), 1.43~1.36(m, 5H), 0.96(t, J=7.3 Hz, 3H); 13C NMR(101 MHz, CDCl3), δ:178.8, 142.3, 138.5, 128.5, 123.4, 122.9, 120.8-110.4(m), 109.7, 43.9, 39.9, 36.9(t, J=20.0 Hz), 29.3, 26.2, 21.9, 20.1, 13.7;19F NMR(376 MHz, CDCl3), δ:-80.8(t, J=12.7 Hz, 3F), -107.5(d, JF-F=341.0 Hz, 1F), -112.3(d, JF-F=341.5 Hz, 1F), -121.8(br, 10F), -122.7(br, 2F), -123.9(br, 2F), -126.2(br, 2F); HRMS m/z(ESI)计算值C25H21F21NO [M+H]+ 750.1283, 实测值:750.1285。
产物3k:1H NMR(400 MHz, CDCl3), δ:7.07(d, J=7.5 Hz, 1H), 6.85(d, J=7.5 Hz, 1H), 6.70(s, 1H), 4.10~3.87(m, 2H), 3.71 (ddt, J=61.0, 14.5, 7.4 Hz, 2H), 2.90~2.67(m, 2H), 2.40(s, 3H), 1.73~1.60(m, 2H), 1.48~1.33(m, 5H), 1.20(t, J=7.1 Hz, 3H), 0.99(t, J=7.3 Hz, 3H); 13C NMR(126 MHz, CDCl3), δ:179.0, 163.6(t, J=32.3 Hz), 143.0, 138.5, 128.0, 123.7, 122.4, 116.7, 114.7(t, J=252.6 Hz), 109.5, 62.8, 44.1(t, J=22.7 Hz), 39.9, 29.3, 25.8, 21.8, 20.1, 13.8, 13.6;19F NMR(376 MHz, CDCl3), δ:-100.8(d, JF-F=266.5 Hz, 1F), -103.6(d, JF-F=266.6 Hz, 1F); HRMS m/z(ESI)计算值C19H26F2NO3 [M+H]+ 354.1876, 实测值:354.1878。
产物3l:1H NMR(400 MHz, CDCl3), δ:7.33(s, 2H), 7.08(s, 1H), 4.25~3.92(m, 2H), 3.89~3.61(m, 2H), 2.96~2.73(m, 2H), 1.75~1.59(m, 2H), 1.50~1.36(m, 5H), 1.22(t, J=7.2 Hz, 3H), 1.00(t, J=7.3 Hz, 3H); 13C NMR(101 MHz, CDCl3), δ:178.4, 163.5, 143.47, 134.9, 130.9(q, J=32.4 Hz), 127.4, 125.0(q, J =278.2 Hz), 118.3, 114.9(t, J=256.7 Hz), 104.9, 63.1, 41.2(t, J=24.8 Hz), 40.1, 31.3, 29.1, 25.7, 20.1, 13.7, 13.6;19F NMR(376 MHz, CDCl3), δ:-62.4(s, 3F), -100.8(d, JF-F=266.5 Hz, 1F), -103.6(d, JF-F=266.6 Hz, 1F); HRMS m/z(ESI)计算值C19H23F5NO3 [M+H]+ 408.1593, 实测值:408.1595。
产物3m:1H NMR(400 MHz, CDCl3), δ:7.38(s, 2H), 7.10(s, 1H), 3.90~3.65(m, 2H), 2.99~2.61(m, 2H), 1.72~1.64(m, 2H), 1.49~1.37(m, 5H), 0.99(t, J=7.3 Hz, 3H); 13C NMR(101 MHz, CDCl3), δ:178.1, 143.2, 135.1, 131.1(q, J=32.5 Hz), 125.4(q, J=278.4 Hz), 124.1, 124.0(q, J=272.2 Hz), 119.6, 105.5, 44.3, 41.0(q, J=28.5 Hz), 40.3, 29.4, 25.5, 20.2, 13.7;HRMS m/z(ESI)计算值C16H18F6NO [M+H]+354.1288, 实测值:354.1290。
1.4 目标产物5的合成
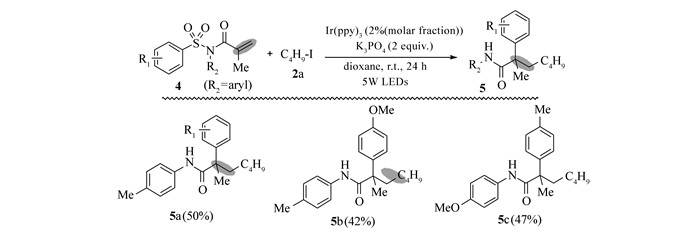
以目标产物5a的合成为例,取原料N-苯甲基-N-甲基丙烯酰基-4-甲基苯磺酰胺88.5 mg(0.3 mmol)溶于2.0 mL二氧六环中,加入fac-Ir(ppy)3 3.6 mg(2.0%(摩尔分数)和K3PO4 127.2 mg(0.6 mmol),再加入C4F9I 207.5 mg(0.6 mmol),并使用N2气保护,在5 W蓝色LED灯照射下反应24 h。经TLC检验反应完全后,经柱层析分离,石油醚-乙酸乙酯梯度洗脱,得到目标产物5a,淡黄色油状液体,产率为50%。
1.5 产物的表征数据
产物5a:黄色油状物; 1H NMR(400 MHz, CDCl3), δ:7.36~7.30(m, 4H), 7.30~7.21(m, 4H), 7.09(dd, J=10.2, 4.3 Hz, 1H), 6.85(s, 1H), 3.21(ddd, J=28.5, 15.7, 12.7 Hz, 1H), 2.89~2.71(m, 1H), 2.37(s, 3H), 1.92(s, 3H); 13C NMR(101 MHz, CDCl3), δ:173.5, 138.0, 137.9, 137.4, 129.9, 129.0, 126.6, 124.6, 120.0, 48.9, 38.1(t, J=19.2 Hz), 23.1, 21.0;19F NMR(376 MHz, CDCl3), δ:-81.0(t, J=9.9 Hz, 3F), -109.5~-110.3(m, 2F), -120.7~-128.3(m, 4F); HRMS m/z(ESI)计算值C21H19F9NO [M+H]+ 472.1317, 实测值:472.1314。
产物5b:黄色油状物; 1H NMR(400 MHz, CDCl3), δ:7.33(d, J=8.2 Hz, 2H), 7.23(dd, J=13.3, 5.9 Hz, 4H), 6.79(dd, J=12.7, 10.8 Hz, 3H), 3.76(s, 3H), 3.29~3.13(m, 1H), 2.87~2.71(m, 1H), 2.37(s, 3H), 1.91(s, 3H); 13C NMR(101 MHz, CDCl3), δ:173.5, 156.7, 138.2, 137.9, 130.5, 129.8, 126.6, 121.9, 114.1, 55.5, 48.7, 38.1(t, J=19.5 Hz), 23.1, 21.0;19F NMR(376 MHz, CDCl3), δ:-80.9(t, J=9.9 Hz, 3F), -109.6~-110.3(m, 2F), -120.7~-128.5(m, 4F); HRMS m/z(ESI)计算值C22H21F9NO2 [M+H]+ 502.1424, 实测值:502.1423。
产物5c:[7]:黄色油状物; 1H NMR(400 MHz, CDCl3), δ: 7.33(d, J=8.2 Hz, 2H), 7.21(s, 2H), 6.84~6.75(m, 4H), 3.76(s, 3H), 3.31~3.11(m, 1H), 2.87~2.67(m, 1H), 2.37(s, 3H), 1.91(s, 3H); 13C NMR(101 MHz, CDCl3), δ:173.5, 156.7, 138.3, 137.9, 130.5, 129.8, 126.6, 122.0, 114.1, 55.5, 48.7, 38.1(t, J=19.5 Hz), 23.1, 21.0;19F NMR(376 MHz, CDCl3), δ:-81.1(t, J=10.0 Hz, 3F), -109.5~-110.4(m, 2F), -120.7~-128.4(m, 4F); HRMS m/z(ESI)计算值C21H19F9NO2 [M+H]+ 488.1267, 实测值:488.1268。
2 结果与讨论
2.1 反应条件探索
以N-正丁基-N-甲基丙烯酰基-4-甲基苯磺酰胺(1a)与全氟碘代丁烷(2a)的环化反应为模板反应探索最佳反应条件(表 1)。首先,对催化剂进行了摸索,令人高兴的是,当使用fac-Ir(ppy)3作为催化剂,在5 W蓝色LED灯照射下,通过串联多氟烷基化/芳基迁移/脱砜化环合反应以78%的产率得到目标产物3a(Entry 1)。接着,考察了其它常见光催化剂(如Ru(bpy)3Cl2,bpy:联吡啶)等,结果发现其催化效果均不如fac-Ir(ppy)3(Entries 2~4)。同时,还对碱进行了筛选,结果表明,K3PO4的效果最好,产率达到51%,而其它无机碱(如K2CO3, Cs2CO3, Na2CO3, KOAc)的效果均不如K3PO4(Entries 5~8)。对溶剂的进一步考察发现二氧六环是最好的溶剂选择,使用其它溶剂(如N, N-二甲基甲酰胺DMF等)产率将会不同程度降低(Entries 9~12)。在接下来的两个控制实验中,我们发现在不使用光催化剂fac-Ir(ppy)3或者在黑暗条件下时无目标产物生成(Entries 13~14),这表明催化剂fac-Ir(ppy)3和光照条件对此反应的进行是必备的。

Entry Initiator Oxidant Solvent Yield of 3a/%b 1 fac-Ir(ppy)3 K3PO4 dioxane 78 2c Ru(bpy)3Cl2 K3PO4 dioxane 39 3c Ru(bpy)3Cl2·6H2O K3PO4 dioxane 32 4c Eosin Y K3PO4 dioxane trace 5 fac-Ir(ppy)3 K2CO3 dioxane 54 6 fac-Ir(ppy)3 Cs2CO3 dioxane 34 7 fac-Ir(ppy)3 Na2CO3 dioxane 15 8 fac-Ir(ppy)3 KOAc dioxane 21 9 fac-Ir(ppy)3 K3PO4 DMF 61 10 fac-Ir(ppy)3 K3PO4 DMSO 19 11 fac-Ir(ppy)3 K3PO4 CH3CN 24 12 fac-Ir(ppy)3 K3PO4 CH2Cl2 trace 13 none K3PO4 dioxane trace 14d fac-Ir(ppy)3 K3PO4 dioxane trace a.Reaction conditions:1a(0.3 mmol), C4F9I(2 stoichiometric number), photocatalyst(2% mole fraction), base(2 stoichiometric number), and solvent(2 mL) were irradiated with a 5 W blue LED at room temperature for 24 h under N2. DMF=N, N-dimethyl formamide, DMSO=dimethyl sulfoxide, Eosin Y=2′, 4′, 5′, 7′-tetrabromofluorescein; b.yield of the isolated product; c.using 5% molar fraction of photocatalyst; d.under darkness. 综上所述,最佳反应条件为:N-正丁基-N-甲基丙烯酰基苯磺酰胺1a(0.3 mmol),K3PO4(2倍化学计量),全氟碘代丁烷2a(2倍化学计量),fac-Ir(ppy)3(2.0%摩尔分数)为光催化剂,二氧六环为溶剂,5 W蓝LED灯为光源,N2气保护,室温下反应24 h。
2.2 反应底物范围
得到最佳反应条件后,我们对反应底物的适用范围进行了考察。实验结果表明,N-烷基苯磺酰胺的N原子上带各种烷基取代基团(如,Bu:丁基, i-Pr:异丙基)时,最佳反应条件对其都表现较好的兼容性。接下来,我们还探究了苯环上取代基对环化反应的影响,令人欣喜的是,在苯环对位上连上各种取代基团时,不论推电子基团或拉电子基团(甲氧基(MeO)、CF3、NO2),均表现有较好的反应活性。接着,我们还对多氟烷基试剂的范围进行了研究,正如我们期待的一样,多种全氟烷基试剂(如C3F7I、C4F9I、C6F13I、C8F17I和C10F21I等)对此脱磺酰环合反应均有较高的活性,能以较好的产率得到相应的全氟烷基化吲哚酮目标产物。此外,CF3I这种氟化试剂也可作为有效氟源用于此反应中,从而在骨架中引入重要三氟甲基基团,只是产率稍微有所降低。芳基磺酰胺的这种串联去磺酰化/全氟烷基化反应,可以将含有各种长度碳链的全氟烷基基团高效地引入有机分子骨架中,这可望成为一种快速、可控的途径去合成各种亲脂的含氟吲哚酮及酰胺,以用于含氟生物活性化合物的筛选。

Entry Substrate Product Yield of 3/% 1 R1=Me, R2=n-Bu, R3=Me, Rf=n-C4F9 3a 78 2 R1=Me, R2=i-Pr, R3=Me, Rf=n-C4F9 3b 60 3 R1=MeO, R2=n-Bu, R3=Me, Rf=n-C4F9 3c 63 4 R1=CF3, R2=n-Bu, R3=Me, Rf=n-C4F9 3d 45 5 R1=NO2, R2=n-Bu, R3=Me, Rf=n-C4F9 3e 41 6 R1=CF3, R2=n-Bu, R3=Me, Rf=n-C3F7 3f 64 7 R1=CF3, R2=n-Bu, R3=Me, Rf=i-C3F7 3g 52 8 R1=Me, R2=n-Bu, R3=Me, Rf=n-C6F13 3h 54 9 R1=Me, R2=n-Bu, R3=Me, Rf=n-C8F17 3i 47 10 R1=Me, R2=n-Bu, R3=Me, Rf=n-C10F21 3j 42 11b R1=Me, R2=n-Bu, R3=Me, Rf=CF2CO2Et 3k 73 12 R1=CF3, R2=n-Bu, R3=Me, Rf= CF2CO2Et 3l 56 13 R1=CF3, R2=n-Bu, R3=Me, Rf=CF3 3m 45 a.Reaction conditions:1(0.3 mmol), Rf-I(2 equiv), Ir(ppy)3(2% molar fraction), K3PO4(2 equiv), and dioxane(2 mL) were irradiated with a 5 W blue LEDs at room temperature for 24 h; b.using BrCF2CO2Et as the fluorine source. 接下来,我们还探究了其它类型可见光诱导下的芳基酰胺的串联脱磺酰/烷基化反应(Scheme 2)。当共轭芳基磺酰胺的N上烷基基团替换为芳基基团时,反应结果发生了较大的变化。此结果与Nevado的报道[6]较为相似,在全氟烷基碘试剂存在下,反应可能是经历了一个自由基串联加成/1, 4芳基迁移/去磺化过程,以中等产率得到α-芳基-β-2-氟/全氟烷基酰胺(5a~5c)。
2.3 反应机理
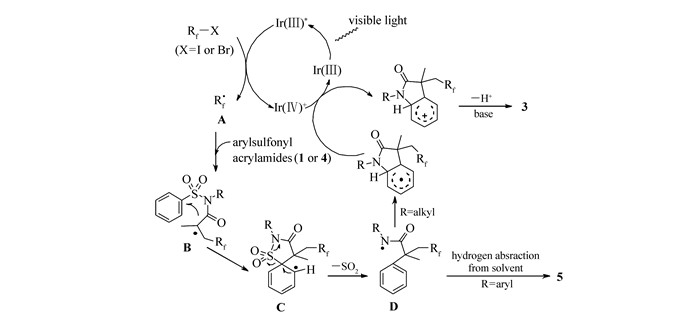
根据以上实验结果及文献报道[3-7],我们提出了可能的反应机理过程(Scheme 3)。首先,在紫外可见光的激发下,光催化剂fac-Ir(kⅢ)(ppy)3在LED蓝光灯照射下转化为其激发态[fac-Ir(Ⅲ)(bpy)3]*,其被全氟碘代丁烷氧化,生成[fac-Ir(Ⅳ)(ppy)3]+以及Rf·自由基A, 接着自由基Rf·对芳基磺酰苯胺(1或4)的C=C双键进行加成,生成中间体B。紧接着B发生分子内自由基环化得到自由基C,然后中间体C发生脱磺酰化得到自由基中间体D[6]。当N原子上取代基为烷基基团时,芳环上的酰基自由基发生分子内环化得到吲哚酮3。然而,当N原子上取代基换成芳基基团时(R=aryl),可能是由于连有芳基基团的N自由基较稳定性的特点[7],自由基D将直接从溶剂夺取氢,从而发生氢化得到产物α-芳基-β-2-氟/全氟烷基酰胺5。
3 结论
本文发展了一种可见光诱导下,共轭苯磺酰胺脱砜环化合成含氟吲哚酮和α-芳基酰胺的方法。此反应以全氟碘代烷烃为氟源,面式-三(2-苯基吡啶)合铱催化N-烷基-N-甲基丙烯酰基苯磺酰胺或N-芳基基-N-甲基丙烯酰基苯磺酰胺发生自由基加成/β-芳基迁移/脱砜环化,快速合成多种含多氟烷基基团的吲哚酮或α-芳基-β-氟烷基酰胺。此方法具有底物适应范围广、反应条件温和(室温),操作简单等特点,为制备全氟吲哚酮和α-芳基酰胺提供了一种途径。
辅助材料(Supporting Information)[目标产物NMR表征图]可以免费从本刊网站(http://yyhx.ciac.jl.cn/)下载。
-
-
[1]
Tomashenko O, Grushin V V. Aromatic Trifluoromethylation with Metal Complexes[J]. Chem Rev, 2011, 111(8): 4475-4521. doi: 10.1021/cr1004293
-
[2]
Filler R Y, Kobayashi. Biomedicinal Aspects of Fluorine Chemistry[M]. Elsevier, Amsterdam, 1982.
-
[3]
Mizuta S, Verhoog S, Engle K M. Catalytic Hydrotrifluoromethylation of Unactivated Alkenes[J]. J Am Chem Soc, 2013, 135(7): 2505-2508. doi: 10.1021/ja401022x
-
[4]
Lin J S, Dong X Y, Li T T. A Dual-Catalytic Strategy to Direct Asymmetric Radical Aminotrifluoromethylation of Alkenes[J]. J Am Chem Soc, 2016, 138(30): 9357-9360. doi: 10.1021/jacs.6b04077
-
[5]
Oh S H, Malpani Y R, Ha N. Vicinal Difunctionalization of Alkenes:Chlorotrifluoromethylation with CF3SO2Cl by Photoredox Catalysis[J]. Org Lett, 2014, 16(5): 1310-1313. doi: 10.1021/ol403716t
-
[6]
Kong W, Casimiro M, Nevado C. Copper-Catalyzed One-Pot Trifluoromethyl-ation/Aryl Migra-tion/Desulfonylation and C(sp2)-N Bond Formation of Conjugated Tosyl[J]. J Am Chem Soc, 2013, 135(39): 14480-14483. doi: 10.1021/ja403954g
-
[7]
Zhang L, Yang C, Xu Z. Difunctionalization of Alkenes via the Visible-Light-Induced Trifluoromethylarylation/1, 4-Aryl Shift/Desulfonylation Cascade Reactions[J]. J Org Chem, 2015, 80(11): 5730-5736. doi: 10.1021/acs.joc.5b00677
-
[8]
Yang F, Klumphu P, Liang Y. Copper-catalyzed Trifluoromethylation of N-Arylacrylamides "On Water" at Room Temperature[J]. Chem Commun, 2014, 50(8): 936-938. doi: 10.1039/C3CC48131J
-
[9]
Itoh Y, Mikami K. Facile Radical Trifluoromethylation of Lithium Enolates[J]. Org Lett, 2005, 7(22): 4883-4885. doi: 10.1021/ol0517574
-
[10]
Qi Q, Shen Q, Lu L. Copper-Mediated Aerobic Fluoroalkylation of Arylboronic Acids with Fluoroalkyl Iodides at Room Temperature[J]. J Am Chem Soc, 2012, 134(15): 6548-6551. doi: 10.1021/ja301705z
-
[11]
Zhang F, Min Q, Zhao H. A General Synthesis of Fluoroalkylated Alkenes by Palladium-Catalyzed Heck-Type Reaction of Fluoroalkyl Bromides[J]. Angew Chem Int Ed, 2015, 54(4): 1270-1274. doi: 10.1002/anie.201409617
-
[12]
Zhang B, Studer A. 6-Perfluoroalkylated Phenanthridines via Radical Perfluoroalkylation of Isonitriles[J]. Org Lett, 2014, 16(15): 3990-3993. doi: 10.1021/ol5018195
-
[13]
Tsuchii K, Imura M, Kamada N. An Efficient Photoinduced Iodoperfluoroalkylation of Carbon-Carbon Unsaturated Compounds with Perfluoroalkyl Iodides[J]. J Org Chem, 2004, 69(20): 6658-6665. doi: 10.1021/jo0495889
-
[14]
Choi Y, Yu C, Kim J. Visible-Light-Induced Arylthiofluoroalkylations of Unactivated Heteroaromatics and Alkenes[J]. Org Lett, 2016, 18(13): 3246-3249. doi: 10.1021/acs.orglett.6b01495
-
[15]
Tang S, Deng Y, Li J. Synthesis of Perfluorinated Isoquinolinediones Through Visible-Light-Induced Cyclization of Alkenes[J]. J Org Chem, 2015, 80(24): 12599-12605. doi: 10.1021/acs.joc.5b01803
-
[1]
-
表 1 优化反应条件筛选
Table 1. Screening of optimal reaction conditions

Entry Initiator Oxidant Solvent Yield of 3a/%b 1 fac-Ir(ppy)3 K3PO4 dioxane 78 2c Ru(bpy)3Cl2 K3PO4 dioxane 39 3c Ru(bpy)3Cl2·6H2O K3PO4 dioxane 32 4c Eosin Y K3PO4 dioxane trace 5 fac-Ir(ppy)3 K2CO3 dioxane 54 6 fac-Ir(ppy)3 Cs2CO3 dioxane 34 7 fac-Ir(ppy)3 Na2CO3 dioxane 15 8 fac-Ir(ppy)3 KOAc dioxane 21 9 fac-Ir(ppy)3 K3PO4 DMF 61 10 fac-Ir(ppy)3 K3PO4 DMSO 19 11 fac-Ir(ppy)3 K3PO4 CH3CN 24 12 fac-Ir(ppy)3 K3PO4 CH2Cl2 trace 13 none K3PO4 dioxane trace 14d fac-Ir(ppy)3 K3PO4 dioxane trace a.Reaction conditions:1a(0.3 mmol), C4F9I(2 stoichiometric number), photocatalyst(2% mole fraction), base(2 stoichiometric number), and solvent(2 mL) were irradiated with a 5 W blue LED at room temperature for 24 h under N2. DMF=N, N-dimethyl formamide, DMSO=dimethyl sulfoxide, Eosin Y=2′, 4′, 5′, 7′-tetrabromofluorescein; b.yield of the isolated product; c.using 5% molar fraction of photocatalyst; d.under darkness. 表 2 环化反应的底物范围a
Table 2. Scope of substrates in cyclizationa

Entry Substrate Product Yield of 3/% 1 R1=Me, R2=n-Bu, R3=Me, Rf=n-C4F9 3a 78 2 R1=Me, R2=i-Pr, R3=Me, Rf=n-C4F9 3b 60 3 R1=MeO, R2=n-Bu, R3=Me, Rf=n-C4F9 3c 63 4 R1=CF3, R2=n-Bu, R3=Me, Rf=n-C4F9 3d 45 5 R1=NO2, R2=n-Bu, R3=Me, Rf=n-C4F9 3e 41 6 R1=CF3, R2=n-Bu, R3=Me, Rf=n-C3F7 3f 64 7 R1=CF3, R2=n-Bu, R3=Me, Rf=i-C3F7 3g 52 8 R1=Me, R2=n-Bu, R3=Me, Rf=n-C6F13 3h 54 9 R1=Me, R2=n-Bu, R3=Me, Rf=n-C8F17 3i 47 10 R1=Me, R2=n-Bu, R3=Me, Rf=n-C10F21 3j 42 11b R1=Me, R2=n-Bu, R3=Me, Rf=CF2CO2Et 3k 73 12 R1=CF3, R2=n-Bu, R3=Me, Rf= CF2CO2Et 3l 56 13 R1=CF3, R2=n-Bu, R3=Me, Rf=CF3 3m 45 a.Reaction conditions:1(0.3 mmol), Rf-I(2 equiv), Ir(ppy)3(2% molar fraction), K3PO4(2 equiv), and dioxane(2 mL) were irradiated with a 5 W blue LEDs at room temperature for 24 h; b.using BrCF2CO2Et as the fluorine source. -

 扫一扫看文章
扫一扫看文章
计量
- PDF下载量: 2
- 文章访问数: 826
- HTML全文浏览量: 172


 下载:
下载:


 下载:
下载:

