
3, 5-二 (吡啶-4-甲氧基) 苯甲酸和芳香二羧酸配体构建的具有3, 4-连接四重和三重穿插的d10金属配位聚合物
-
关键词:
- 配位聚合物
- / 3, 5-二 (吡啶-4-甲氧基) 苯甲酸
- / 晶体结构
- / 荧光性质
English
Four-and Three-Fold Interpenetrated (3, 4)-Connected d10 Metal Coordination Polymers Constructed by 3, 5-Bis (pyridin-4-ylmethoxy) benzoic Acid and Aromatic Dicarboxylic Acid Ligands
-
0 Introduction
Crystal engineering of coordination polymers (CPs), which involves self-assembly of organic ligands with appropriate functional groups and metal ions with specific directionality and functionality, is one of the facile routes to produce materials of technological importance[1]. The metal-ligand coordination bonds have been widely exploited in organizing molecular building blocks into diverse supramolecular architectures, making use of the strength of coordination bonds and directionality associated with metal ions[2]. Coordina-tion polymers have attracted a great of extensive interest not only for their potential applications in adsorption, nonlinear optics, magnetism, photolumine-scence, catalysis, and electrical conductivity, but also for their intriguing variety of architectures and fascin-ating topologies[3]. A number of topological types are applied in various CPs, because of its ability of simplifying complicated frameworks and the instru-ctive role in the rational design of some predicted functional materials[4]. Of particular interest are inter-penetrating frameworks[5]. These species can be reg-arded as infinite ordered polycatenanes and are characterized by the presence of two or more independent networks that cannot be separated in a topological sense without breaking of bonds. In the past decade, considerable effort has been invested in interpenetrating CPs because of their unique advantages in enhancing stability, specific surface area, gas sorption, and molecular dynamics, as well as their general structural aesthetics [6].
The long-flexible ligand may be a good candidate as a unique structural motif to produce interpene-trating architectures with interesting topologies and useful functional properties[7]. Hence we selected 3, 5-bis (pyridin-4-ylmethoxy) benzoic acid (HL) as primary ligand, because it contains a rigid spacer of phenyl ring and two freely rotating pyridyl arms, which may cause the planes of the pyridyl rings to rotate with respect to the plane of the central phenyl ring. On the other hand, d10 metal (Cd (Ⅱ), Zn (Ⅱ)) compounds have attracted extensive interest in recent years because of photoluminescent properties[8]. With this aim of under-standing the coordination chemistry of HL[9-10] and preparing new materials with interpenetrating networks and excellent physical properties, we have recently engaged in the research of the CPs with HL ligand[10]. In this paper, we describe the preparations, crystal structures, and photoluminescent properties of four CPs based on HL and dicarboxylic acid ligands, namely, [Zn2L2 (tdc)]n (1), {[Zn4L4(hbdc)2]·H2O}n (2), {[Zn2L2(bdtc)]·H2O}n (3) and {[Cd2L2 (bdtc) H2O]·0.5H2O}n (4).
1 Experimental
1.1 Materials and physical measurements
The ligands and other reagents were purchased from Jinan Camolai Trading Company and used without further purification. The elemental analyses of C, H and N were performed on a Perkin-Elmer 2400 Ⅱ elemental analyzer. IR spectrum was measured in KBr pellets on a Nicolet 5DX FT-IR spectrometer. The thermogravimetric measurement was performed on preweighed samples in an oxygen stream using a Netzsch STA449C apparatus with a heating rate of 10 ℃·min-1. The crystal data were acquired on a Bruker APEX Ⅱ diffractometer. Powder X-ray diffraction data were obtained using a Philips PW3040/60 auto-mated powder diffractometer, using Cu Kα radiation (λ=0.154 2 nm) with a 2θ range of 5°~50 °. The lumi-nescence spectra were performed on a HITACHIF-2500 fluorescence spectrometer in solid state at room temperature.
1.2 Synthesis of [Zn2L2(tdc)]n (1)
A mixture of ZnSO4·2H2O (0.20 mmol, 0.058 g), HL (0.10 mmol, 0.034 g), H2tdc (0.05 mmol, 0.013 g) and NaOH (0.15 mmol, 0.006 g) in H2O (14 mL) was sealed in a 25 mL Teon-lined stainless steel container, which was heated at 160 ℃ for 72 h and then cooled down to room temperature at a rate of 5 ℃·h-1. Yellow crystals of 1 were collected and washed with distilled water and dried in air to give the product with 27.4% yield (based on HL). Anal. Calcd. for C44H32N4O12SZn2 (%): C, 54.40; H, 3.32; N, 5.77. Found (%): C, 54.18; H, 3.39; N, 5.86. IR (KBr pellet, cm-1): 3 444, 3 086, 2 930, 2 366, 1 631, 1 603, 1 419, 1 376, 1 235, 1 150, 1 065, 868, 797.
1.3 Syntheses of {[Zn4L4(Hhdc)2]·H2O}n (2) and {[Zn2L2(Hhdc)]·H2O}n (3)
A mixture of ZnSO4·2H2O (0.20 mmol, 0.058 g), HL (0.10 mmol, 0.034 g), H3hdc (0.10 mmol, 0.018 g) and NaOH (0.30 mmol, 0.012 g) in H2O (14 mL) was sealed in a 25 mL Teflon-lined stainless steel container, which was heated at 160 ℃ for 72 h and then cooled down to room temperature at a rate of 5 ℃·h-1. The containing pale yellow blocklike crystals of 2 and yellow platelike crystals of 3 were obtained. The yield of 2 is about 3.0% (based on HL). Anal. Calcd. for C92H72N8O27Zn4(%): C, 55.78; H, 3.56; N, 5.66. Found (%): C, 55.70; H, 3.62; N, 5.74. IR (KBr pellet, cm-1): 3 429, 2 943, 1 640, 1 560, 1 367, 1 249, 1 161, 1 073, 1 028, 910, 792. The yield of 3 is 30.1% (based on HL). Anal. Calcd. for. C46H36N4O14Zn2(%): C, 55.72; H, 3.66; N, 5.65. Found (%): C, 55.39; H, 3.62; N, 5.52. IR (KBr pellet, cm-1): 3 458, 3 075, 2 943, 2 354, 1 617, 1 581, 1 381, 1 279, 1 175, 1 057, 1 014, 778, 719, 497.
1.4 Synthesis of {[Cd2L2(bdtc)(H2O)]·0.5H2O}n (4)
A mixture of Cd (CH3COO)2·2H2O (0.20 mmol, 0.053 g), HL (0.10 mmol, 0.034 g), H2bdtc (0.05 mmol, 0.013 g) and NaOH (0.10 mmol, 0.004 g) in H2O (14 mL) was sealed in a 25 mL Teflon-lined stainless steel container, which was heated at 160 ℃ for 72 h and then cooled down to room temperature at a rate of 5 ℃·h-1. Yellow crystals of 4 were collected and washed with distilled water and dried in air to give the product with 11.8% yield (based on HL). Anal. Calcd. for C48H44Cd2N4O15S2(%): C, 47.81; H, 3.68; N, 4.65. Found (%): C, 47.86; H, 3.72; N, 4.61. IR (KBr pellet, cm-1): 3 430, 2 970, 2 919, 2 368, 1 613, 1 551, 1 480, 1 377, 1 235, 1 163, 1 102, 898, 796, 673.
1.5 X-ray crystallography study
Data collection of 1~4 was carried out on a Bruker APEX Ⅱ diffractometer equipped with a graphite-monochromatized Mo Kα radiation (λ=0.071 073 nm) at 296(2) K. Data intensity was corrected by Lorentz-polarization factors and empirical absorption. The structure was solved by the direct methods and refined with the full-matrix least-squares refinements technique based on F2. The anisotropic displacement parameters were applied to all non-hydrogen atoms. The hydrogen atoms were assigned with isotropic displacement factors and included in the final refinement cycles using geometrical restrains. All calculations were performed with SHELXS-97 and SHELXTL-97 program packages[11]. The detailed crys-tallographic data and structure refinement parameters are summarized in Table 1. Selected bond lengths and angles are given in Table 2.
Complex 1 2 3 4 Empirical formula C44H32N4O12SZn2 C92H72N8O27Zn4 C46H36N4O14Zn2 C48H44Cd2N4O15S2 Formula weight 971.54 1 983.06 999.52 1 205.79 Crystal system Monoclinic Monoclinic Monoclinic Triclinic Space group P21/c P2 /c P21 /c P1 a / nm 1.004 50(4) 1.347 87(3) 1.358 17(5) 0.808 78(2) b/ nm 2.133 30(9) 1.101 06(2) 1.510 87(6) 1.186 59(3) c / nm 1.075 02(4) 2.011 82(3) 2.335 31(9) 1.404 32(4) α/(°) 71.917(2) β/(°) 111.749(2) 131.200(1) 104.802(2) 86.625 0(10) γ/(°) 82.307 0(10) V/nm3 2.139 68(15) 2.246 49(7) 4.633 1(3) 1.269 40(6) Z 2 1 4 1 Dc/ (g·cm-3) 1.508 1.464 1.433 1.577 μ/ mm-1 1.238 1.138 1.106 0.990 F(000) 992 1 014 2 048 608 θmin, θmax / (°) 1.91, 27.55 1.85, 27.54 1.55, 27.51 1.53, 27.62 Reflections collected 34 136 18 261 71 272 19 824 Unique reflections (Rint) 4 910 (0.055 2) 5 160 (0.049 8) 10 590 (0.09 60) 5 779 (0.034 2) Data with I>2σ(I) 4 910 5 160 5 411 4 292 Parameters refined 307 299 594 334 R, wR [I>2σ(I)] 0.041 3, 0.101 6 0.053 9, 0.149 8 0.062 8, 0.179 3 0.038 9, 0.120 1 R, wR (all data) 0.066 7, 0.113 4 0.101 7, 0.1642 0.139 2, 0.219 6 0.046 9, 0.125 6 Goodness-of-fit on F2 1.040 0.991 0.978 1.077 (△ρ)max, (△ρ)min/ (e·nm-3) 404, -358 1 050, 279 892, -431 1 271, -512 Table 1. Crystal data and structure parameters for compounds 1~4Complex 1 Zn (1)-O (5) 0.192 0(3) Zn (1)-O (1) 0.193 52(19) Zn (1)-N (1)#1 0.202 1(2) Zn (1)-N (2)#2 0.207 2(2) O (5)-Zn (1)-O (1) 110.76(11) O (5)-Zn (1)-N (1)#1 117.54(12) O (1)-Zn (1)-N (1)#1 115.97(10) O (5)-Zn (1)-N (2)#2 111.25(11) O (1)-Zn (1)-N (2)#2 95.93(9) N (1)#1-Zn (1)-N (2)#2 102.75(9) Complex 2 Zn (1)-O (1) 0.192 9(3) Zn (1)-O (6) 0.196 4(3) Zn (1)-N (1) #1 0.205 5(3) Zn (1)-N (2)#2 0.206 9(3) Zn (1)-O (7) 0.258 1(3) O (1)-Zn (1)-0(6) 133.61(13) O (1)-Zn (1)-N (1)#1 98.21(13) 0(6)-Zn (1)-N (1)#1 111.56(13) O (1)-Zn (1)-N (2)#2 114.07(14) O (6)-Zn (1)-N (2)#2 95.69(13) N (1)#1-Zn (1)-N (2)#2 99.50(13) O (6)-Zn (1)-O (7) 54.88(11) N (1)-Zn (1)-O (7)#1 89.17(13) N (2)-Zn (1)-O (7)#2 150.25(12) Complex 3 Zn (1)-O (1) 0.200 9(4) Zn (1)-O (2) 0.252 1(4) Zn (1)-O (11) 0.205 8(4) Zn (1)-N (1)#1 0.205 0(4) Zn (1)-O (10) 0.240 4(5) Zn (1)-N (2)#2 0.209 0(4) Zn (2)-O (13) 0.191 8(3) Zn (2)-N (3)#1 0.202 7(4) Zn (2)-O (6) 0.193 5(3) Zn (2)-N (4)#3 0.203 6(4) O (1)-Zn (1)-N (1)#1 96.36(17) O (1)-Zn (1)-0(11) 134.58(16) N (1)#1-Zn (1)-O (11) 117.26(16) O (1)-Zn (1)-N (2)#2 111.74(16) N (1)#1-Zn (1)-N (2)#2 101.94(16) O (11)-Zn (1)-N (2)#2 91.26(16) O (1)-Zn (1)-O (10) 94.00(15) N (1)#1-Zn (1)-O (10) 92.46(15) O (11)-Zn (1)-O (10) 57.30(14) N (2)#2-Zn (1)-O (10) 148.52(15) O (13)-Zn (2)-O (6) 128.19(17) O (13)-Zn (2)-N (3)#1 106.79(18) O (6)-Zn (2)-N (3)#1 99.00(17) O (13)-Zn (2)-N (4)#3 97.81(16) O (6)-Zn (2)-N (4)#3 116.47(16) N (3)#1-Zn (2)-N (4)#3 107.23(18) Complex 4 Cd (1)-N (2)#1 0.225 4(3) Cd (1)-O (2) 0.226 0(3) Cd (1)-O (6) 0.229 3(3) Cd (1)-N (1)#2 0.232 4(3) Cd (1)-O (1W) 0.238 5(3) Cd (1)-O (1) 0.247 6(3) N (2)#1-Cd (1)-O (2) 150.06(11) N (2)#1-Cd (1)-O (6) 99.20(12) 0(2)-Cd (1)-O (6) 86.53(11) N (2)#1-Cd (1)-N (1)#2 110.59(12) O (2)-Cd (1)-N (1)#2 98.78(11) O (6)-Cd (1)-N (1)#2 89.27(11) N (2)#1-Cd (1)-O (1W) 84.09(11) O (2)-Cd (1)-O (1W) 92.60(10) O (6)-Cd (1)-O (1W) 174.82(9) N (1)#2-Cd (1)-O (1W) 84.09(11) N (2)#1-Cd (1)-O (1) 95.00(10) O (2)-Cd (1)-O (1) 55.21(9) O (6)-Cd (1)-O (1) 92.92(10) N (1)#2-Cd (1)-O (1) 153.64(11) O (1W)-Cd (1)-O (1) 90.77(10) Symmetry codes: #1:-x+1, y+1/2, -z+3/2, #2 x-1, -y+1/2, z+1/2, #3-x+1, y-1/2, -z+3/2, #4 x+1, -y+1/2, z-1/2 for 1; #1 x-1, y, z, #2-x-1, y+1, -z-1/2 for 2; #1 x-1, y, z, #2 x, -y+1/2, z+1/2, #3 x, -y-3/2, z+1/2 for 3; #1 x-1, y+1, z, #2 x, y, z+1 for 4. Table 2. Selected bond lengths (nm) and angles (°) for 1~4CCDC: 852424, 1; 872035, 2; 887738, 3; 872037, 4.
2 Results and discussion
Four new 3D metal (Ⅱ) CPs have been synthesized by hydrothermal reactions of d10 metal (Ⅱ) salts, 3, 5-bis (pyridin-4-ylmethoxy) benzoic acid (HL) and dicarbo-xylic acid ligands. In the process of synthesis of 3, a by-product 2 was co-precipitated unexpectedly, and there are some differences of the crystal shape and color between 2 and 3. However, the amount of 2 difficultly resynthesized was not enough to study its other properties.
2.1 Structure of [Zn2L2(tdc)]n (1)
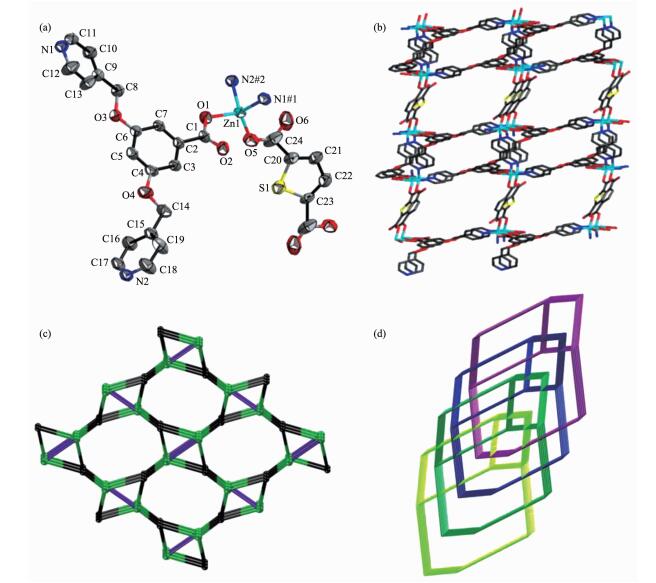
Single-crystal X-ray diffraction analysis shows that 1 crystallizes in monoclinic with P21/c space group. The asymmetric unit of 1 consists of one Zn (Ⅱ) ion, one L- ligand and half tdc2- ligand. As shown in Fig. 1a, each Zn (Ⅱ) ion is four-coordinated with a distorted tetrahedral geometry supplied by two carboxylate oxygen atoms from one L- ligand and one tdc2- ligand, two nitrogen atoms from two L- ligands. As shown in Fig. 1b, the L- ligand connects three Zn (Ⅱ) ions together by two pyridyl-N atoms and one carboxylate oxygen atom to generate a 2D bilayer structure, and then the tdc2- ligands locate up and down the layer in exo-bidentate mode to form the final 3D network.
Topologically, each Zn (Ⅱ) ion is attached to three L- ligands and one tdc2- ligand, which can be considered as a 4-connected node; each L- ligand connects three Zn (Ⅱ) ions and can be considered as a 3-connected node; while each tdc2- ligand can be seen as a linear linker between two Zn (Ⅱ) ions. Therefore, the overall network can be described as a (3, 4)-connected dmc network with point symbol of (4.82)(4.85) (Fig. 1c). Because of the spacious nature of the single network, the potential voids are filled via mutual interpenetration of identical 3D frameworks, generating a 4-fold interpenetrating architecture (Fig. 1d).
2.2 Structure of {[Zn4L4(Hhdc)2]·H2O}n (2)
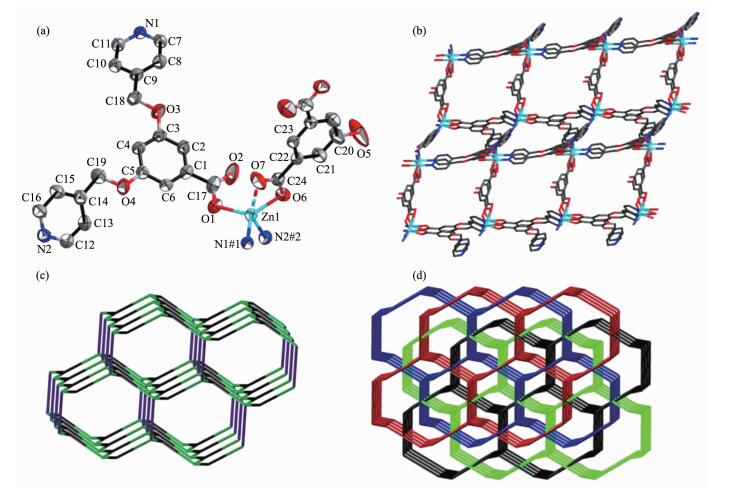
Crystallographic analysis reveals that 2 crystall-izes in the monoclinic with space group P2/c. The asymmetric unit of 2 consists of one Zn (Ⅱ) ion, one L- ligand, half Hhdc2- ligand and a quarter of lattice H2O molecule. As shown in Fig. 2a, each Zn (Ⅱ) ion is four-coordinated with a distorted tetrahedral geometry supplied by two pyridyl-N atoms from two L- ligands, two carboxylic oxygen atoms from one L- ligand and one Hhdc2- ligand. It is worthy to note that the distance of Zn (1)…O (7) is 0.258 1(3) nm, which is a nonnegligible week interaction, and the Zn (Ⅱ) ion can be considered as a five coordinated, disorted square pyramidal geometry. As shown in Fig. 2b, the L- ligand connects three Zn (Ⅱ) ions together by two pyridyl-N atoms and one carboxylate oxygen atom to generate a 2D layer structure, and then the Hhdc2- ligands locate up and down the layer in exo-bidentate mode to form the final 3D network.
Each Zn (Ⅱ) ion is attached to three L- ligands and one Hhdc2- ligand to represent a 4-connected node; each L- ligand presents a 3-connected node; and each Hhdc2- ligand can be seen as a linear linker between two Zn (Ⅱ) ions. 2 possesses a 3D (3, 4)-connected fsc net with point symbol of (63)(65.8) topo-logy (Fig. 2c). The identical 3D frameworks interlock each other, generating a 4-fold interpenetrating archi-tecture (Fig. 2d).
2.3 Structure of {[Zn2L2(Hhdc)]·H2O}n (3)
Crystallographic analysis reveals that 3 crystall-izes in the monoclinic with space group P21/c. The asymmetric unit of 3 consists of two Zn (Ⅱ) ions, two L- ligands, one Hhdc2- ligand and one lattice H2O molecule. As shown in Fig. 3a, Zn (1) is connected by four carboxylic oxygen atoms from one L- ligand and one Hhdc2- ligand, two pyridyl-N atoms from two L- ligands in a disorted octahedral geometry, and Zn (2) is connected by two carboxylic oxygen atoms from one L- ligand and one Hhdc2- ligand, two pyridyl-N atoms from two L- ligands in a distorted tetrahedral coordina-tion geometry. As shown in Fig. 3b, the L- ligands connect three Zn (Ⅱ) ions together to generate a 2D layer structure, and then the Hhdc2- ligands locate up and down the layers in exo-bidentate mode to form the final 3D network.
Each Zn (Ⅱ) ion is attached to three L- ligands and one Hhdc2- ligand to represent a 4-connected node; each L- ligand presents a 3-connected node; and each Hhdc2- ligand can be seen as a linear linker between two Zn (Ⅱ) ions. 3 possesses a 3D (3, 4)-connected fsc net with the (63)(65.8) topology and a 4-fold interpenetrating architecture.
2.4 Structure of {[Cd2L2(bdtc)(H2O)]·0.5H2O}n (4)
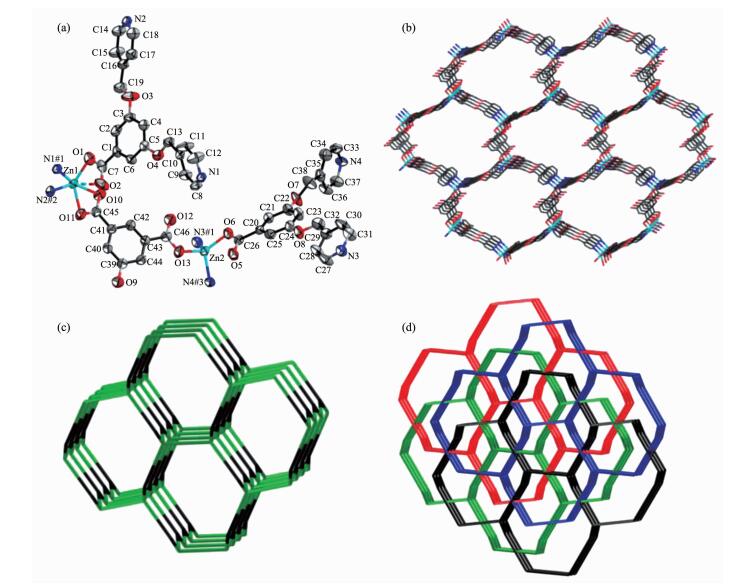
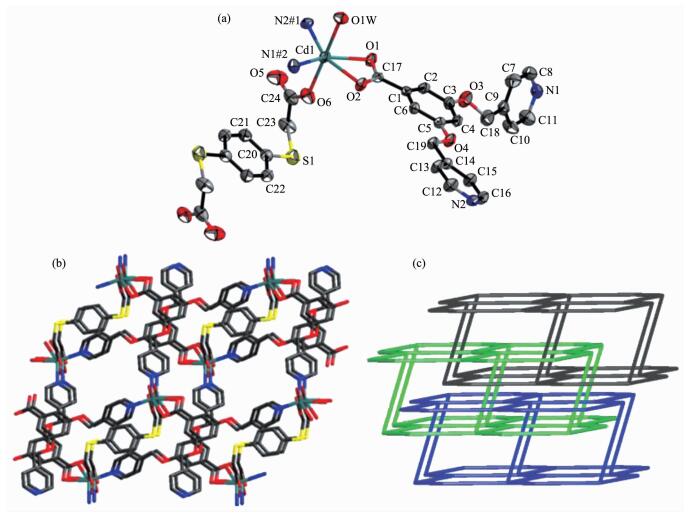
Crystallographic analysis reveals that 4 crysta-llizes in the triclinic space group P1. The asymmetric unit of 4 consists of one Cd (Ⅱ) ion, one L- ligand, half bdtc2- ligand, half coordinated water molecule and quarter lattice water molecule. As shown in Fig. 4a, each Cd (Ⅱ) ion is six-coordinated with a distorted octahedral geometry supplied by two pyridyl-N atoms from two L- ligands, three carboxylic oxygen atoms from one L- ligand and one bdtc2- ligand, and one water molecule. As shown in Fig. 4b, the L- ligand connects three Cd (Ⅱ) ions together by two pyridyl-N atoms and two carboxylate oxygen atoms, the bdtc2- ligand connect two Cd (Ⅱ) ions together by two carboxylate oxygen atoms to generate a 2D (3, 4)-conn-ected layer structure.
The topology analysis suggests the (3, 4)-connected net with the (63)(66) topology, and each 2D layer is filled via mutual interpenetration to generate a 3-fold interpenetrating architecture (Fig. 4c). Simultan-eously, the adjacent layers are linked each other by hydrogen bonds (O (1W)-H (1WB)…O (5)#3 0.276 1(5) nm, O (3W)-H (1WA)…O5#4 0.276 8(13) nm; Symmetry codes: #3: x+1, y, z, 4#:-x, 1-y, 2-z) to complete the finally 3D supramolecular network.
2.5 PXRD analysis and thermal property
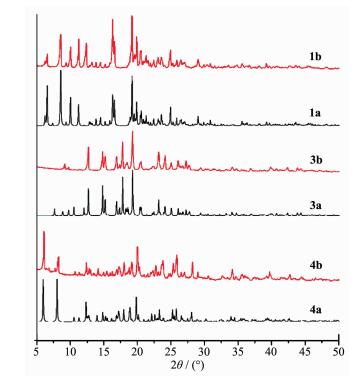
Powder X-ray diffraction (PXRD) analysis of 1, 3, 4 has been performed at room temperature. The PXRD patterns are in reasonably good agreement with the calculated ones (Fig. 5), confirming the phase purity of the as-synthesized products.
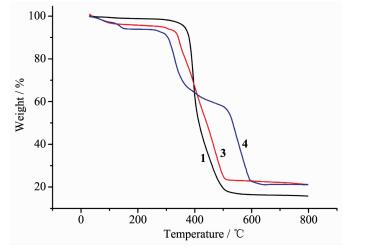
Thermogravimetric analyses (TGA) were carried out under air atmosphere to examine the thermal stability of complexes 1, 3 and 4 and the results are shown in Fig. 6. The TGA curve of 1 shows the framework collapsed at the temperature range of 305~510 ℃. For 3, the first weight loss of 1.88 % occurred in the temperature range of 70~120 ℃, due to the release of coordinated water molecules (Calcd. 1.80%), and the decomposition of the residue occurs at the range of 320~510 ℃. The TGA curve of 4 shows the first weight loss of 2.51% occurred in the temperature range of 100~145 ℃, due to the release of water molecules (Calcd. 2.24%), and the decomposition of the residue occurs at the range of 275~330 ℃, which may be caused by the complete decomposition of bdtc in 4 (Obsd. 19.87%, Calcd. 19.24%). A rapid weight loss can be detected from 500~600 ℃, which may be attributed to the complete decomposition of the organic ligands.
2.6 Photoluminescent properties
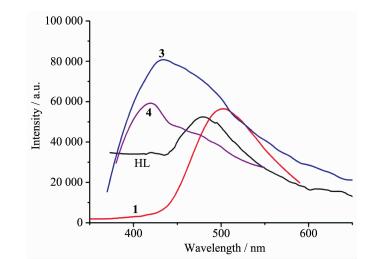
Taking the excellent luminescent properties of Zn (Ⅱ) and Cd (Ⅱ) into account, the solid-state lumin-escent spectra of HL, complexes 1, 3 and 4 have been studied at room temperature (Fig. 7). 1 shows broad fluorescent emission spectra from 450 to 600 nm and the emission maxima at 504 nm (λex=300 nm). 3 shows broad fluorescent emission spectra from 400 to 600 nm and the emission maxima at 450 nm (λex=360 nm), while 4 presents broad fluorescent emission spectra from 400 to 550 nm and the emission maxima at 452 nm (λex=362 nm). In order to understand the nature of the emission, we analyzed the photolumin-escence property of free HL ligand and found that the strongest emission peak at 474 nm (λex=363 nm). In comparison with that of HL, the maximum emission wavelengths of 1 occurs slightly redshifted, 3 and 4 occur slightly blueshifted, which are probably due to L-and auxiliary ligands to metal charge transitions. The enhancement of the intensity of luminescence in the CPs may be attributed to the rigidity of them. The complex enhances the "rigidity" of the ligand and thus reduces the loss of energy through a radiationless pathway[12].
3 Conclusions
In summary, complexes 1~4 display four-and three-fold interpenetrated (3, 4)-connected coordination polymers. The interpenetration effectively reduced the solvent voids, in which clathrate water molecules reside. The results show that the flexible ligand can take different modes to meet the coordination nature of the metal cations, and the ligands, the metal center, and the reaction conditions have great influence on the structure of the final assembly. The long-flexible ligand with rigid spacers is much more conducive to form interpenetrated framework. The complexes with spacious voids in their frameworks favour interpenetrated crystal forms. The luminescent spectra show that 1, 3 and 4 have different luminescent properties, both the auxiliary ligands and metal centers will influence the intensity and shift of luminescence.
-
-
[1]
(a) Leong W L, Vitta J J. Chem. Rev. , 2011, 111: 688-764
(b) Moulton B, Zaworotko M. Chem. Rev. , 2001, 101: 1629-1658
(c) Zhao D, Timmons D J, Yuan D, et al. Acc. Chem. Res. , 2011, 44: 123-133 -
[2]
(a) Hong M C, Chen L. Design and Construction of Coordina-tion Polymers. Hoboken, New Jersey: John Wiley & Sons, Inc. , 2009.
(b) Batten S R, Neville S M, Turner D R. Coordination Polymers: Design, Analysis and Application. Cambridge UK: Royal Society of Chemistry, 2009. -
[3]
(a) Lin Z J, Lu J, Hong M C, et al. Chem. Soc. Rev. , 2014, 43: 5867-5895
(b) Stock N, Biswas S. Chem. Rev. , 2012, 112: 933-969
(c) Li J R, Sculley J, Zhou H C. Chem. Rev. , 2012, 112: 869-932
(d) Moon H R, Lim D W, Suh M P. Chem. Soc. Rev. , 2013, 42: 1807-1824 (e) Chaemchuen S, Kabir N A, Zhou K, et al. Chem. Soc. Rev. , 2013, 42: 9304-9332 -
[4]
(a) O'Keeffe M, Hyde S T. Zeolites, 1997, 19: 370-374
(b) O'Keeffe M. Nature, 1999, 400: 617
(c) Ockwig N W, Friedrichs O D, O'Keeffe M, et al. Acc. Chem. Res. , 2005, 38: 176-182 -
[5]
(a) Wang R, Wang Z, Xu Y, et al. Inorg. Chem. , 2014, 53: 7086-7088
(b) Li F, Clegg J K, Lindoy L F, et al. Nat. Commun. , 2001, 205: 1208
(c) Wang R, Zhang M, Liu X, et al. Inorg. Chem. , 2015, 54: 6084-6086 -
[6]
(a) Wu H, Yang J, Su Z M, et al. J. Am. Chem. Soc. , 2011, 133: 11406-11409
(b) Kitagawa S, Matsuda R. Coord. Chem. Rev. , 2007, 251: 2490-2509 -
[7]
Schneemann A, Bon V, Schwedler I, et al. Chem. Soc. Rev., 2014, 43:6062-6096 doi: 10.1039/C4CS00101J
-
[8]
(a) Wang X L, Qin C, Wu S X, et al. Angew. Chem. Int. Ed. , 2009, 48: 5291-5295
(b) Zheng S L, Yang J H, Yu X L, et al. Inorg. Chem. , 2004, 43: 830-836
(c) Zhang L Y, Zhang J P, Lin Y Y, et al. Cryst. Growth Des. , 2006, 6: 1684-1689
(d) Rather B, Moulton B, Walsh R D B, et al. Chem. Commun. , 2002: 694-695 -
[9]
(a) Xu G J, Zhao Y H, Shao K Z, et al. CrystEngComm, 2009, 11: 1842-1848
(b) Xu G J, Zhao Y H, Shao K Z, et al. Polyhedron, 2009, 28: 3155-3163
(c) Xu G J, Zhao Y H, Shao K Z, et al. Inorg. Chem. Commun. , 2008, 11: 1181-1183
(d) Xu G J, Zhao Y H, Shao K Z, et al. Inorg. Chem. Commun. , 2010, 13: 932-934
(e) Xu G J, Zhao Y H, Shao K Z, et al. Inorg. Chem. Commun. , 2009, 12: 1024-1026
(f) Xu G J, Zhao Y H, Shao K Z, et al. Inorg. Chem. Commun. , 2009, 12: 969-971 -
[10]
(a) HAN Min-Min(韩敏敏), CHEN Xiao(陈晓), ZHANG Yi-Ping(张依萍), et al. Chinese J. Inorg. Chem.(无机化学学报) , 2014, 30: 1653-1659
(b) Cao K L, Zhang Y P, Cai Y N, et al. J. Solid State Chem. , 2014, 215: 34-42 -
[11]
(a) Sheldrick G M. SHELXS 97, University of Göttingen, Germany, 1997.
(b) Sheldrick G M. SHELXTL 97, University of Göttingen, Germany, 1997. -
[12]
He Y H, Feng Y L, Lan Y Z, et al. Cryst. Growth Des., 2008, 8:3586-1694 doi: 10.1021/cg8000398
-
[1]
-
Figure 1 (a) Coordination environment of Zn (Ⅱ) in 1; (b) 3D coordination framework; (c) Schematic representation of the (3, 4)-connected framework; (d) Schematic representation of the 4-fold interpenetrating net
Thermal ellipsoids are shown at the 50% probability level in (a); Symmetry codes: #1:-x+1, y+1/2, -z+3/2, #2: x-1, -y+1/2, z+1/2
Figure 2 (a) Coordination environment of Zn (Ⅱ) in 2; (b) 3D coordination framework; (c) Schematic representation of the (3, 4)-connected framework; (d) Schematic representation of the 4-fold interpenetrating net
Thermal ellipsoids are shown at the 50% probability level in (a); Symmetry codes: #1: x-1, y, z; #2:-x+1/2, y+1, -z-1/2
Figure 3 (a) Coordination environment of Zn (Ⅱ) in 3; (b) 3D coordination framework; (c) Schematic representation of the (3, 4)-connected framework; (d) Schematic representation of the 4-fold interpenetrating net
Thermal ellipsoids are shown at the 50% probability level in (a); Symmetry codes: #1: x-1, y, z; #2: x, -y+1/2, z+1/2; #3: x, -y-3/2, z+1/2
Table 1. Crystal data and structure parameters for compounds 1~4
Complex 1 2 3 4 Empirical formula C44H32N4O12SZn2 C92H72N8O27Zn4 C46H36N4O14Zn2 C48H44Cd2N4O15S2 Formula weight 971.54 1 983.06 999.52 1 205.79 Crystal system Monoclinic Monoclinic Monoclinic Triclinic Space group P21/c P2 /c P21 /c P1 a / nm 1.004 50(4) 1.347 87(3) 1.358 17(5) 0.808 78(2) b/ nm 2.133 30(9) 1.101 06(2) 1.510 87(6) 1.186 59(3) c / nm 1.075 02(4) 2.011 82(3) 2.335 31(9) 1.404 32(4) α/(°) 71.917(2) β/(°) 111.749(2) 131.200(1) 104.802(2) 86.625 0(10) γ/(°) 82.307 0(10) V/nm3 2.139 68(15) 2.246 49(7) 4.633 1(3) 1.269 40(6) Z 2 1 4 1 Dc/ (g·cm-3) 1.508 1.464 1.433 1.577 μ/ mm-1 1.238 1.138 1.106 0.990 F(000) 992 1 014 2 048 608 θmin, θmax / (°) 1.91, 27.55 1.85, 27.54 1.55, 27.51 1.53, 27.62 Reflections collected 34 136 18 261 71 272 19 824 Unique reflections (Rint) 4 910 (0.055 2) 5 160 (0.049 8) 10 590 (0.09 60) 5 779 (0.034 2) Data with I>2σ(I) 4 910 5 160 5 411 4 292 Parameters refined 307 299 594 334 R, wR [I>2σ(I)] 0.041 3, 0.101 6 0.053 9, 0.149 8 0.062 8, 0.179 3 0.038 9, 0.120 1 R, wR (all data) 0.066 7, 0.113 4 0.101 7, 0.1642 0.139 2, 0.219 6 0.046 9, 0.125 6 Goodness-of-fit on F2 1.040 0.991 0.978 1.077 (△ρ)max, (△ρ)min/ (e·nm-3) 404, -358 1 050, 279 892, -431 1 271, -512 Table 2. Selected bond lengths (nm) and angles (°) for 1~4
Complex 1 Zn (1)-O (5) 0.192 0(3) Zn (1)-O (1) 0.193 52(19) Zn (1)-N (1)#1 0.202 1(2) Zn (1)-N (2)#2 0.207 2(2) O (5)-Zn (1)-O (1) 110.76(11) O (5)-Zn (1)-N (1)#1 117.54(12) O (1)-Zn (1)-N (1)#1 115.97(10) O (5)-Zn (1)-N (2)#2 111.25(11) O (1)-Zn (1)-N (2)#2 95.93(9) N (1)#1-Zn (1)-N (2)#2 102.75(9) Complex 2 Zn (1)-O (1) 0.192 9(3) Zn (1)-O (6) 0.196 4(3) Zn (1)-N (1) #1 0.205 5(3) Zn (1)-N (2)#2 0.206 9(3) Zn (1)-O (7) 0.258 1(3) O (1)-Zn (1)-0(6) 133.61(13) O (1)-Zn (1)-N (1)#1 98.21(13) 0(6)-Zn (1)-N (1)#1 111.56(13) O (1)-Zn (1)-N (2)#2 114.07(14) O (6)-Zn (1)-N (2)#2 95.69(13) N (1)#1-Zn (1)-N (2)#2 99.50(13) O (6)-Zn (1)-O (7) 54.88(11) N (1)-Zn (1)-O (7)#1 89.17(13) N (2)-Zn (1)-O (7)#2 150.25(12) Complex 3 Zn (1)-O (1) 0.200 9(4) Zn (1)-O (2) 0.252 1(4) Zn (1)-O (11) 0.205 8(4) Zn (1)-N (1)#1 0.205 0(4) Zn (1)-O (10) 0.240 4(5) Zn (1)-N (2)#2 0.209 0(4) Zn (2)-O (13) 0.191 8(3) Zn (2)-N (3)#1 0.202 7(4) Zn (2)-O (6) 0.193 5(3) Zn (2)-N (4)#3 0.203 6(4) O (1)-Zn (1)-N (1)#1 96.36(17) O (1)-Zn (1)-0(11) 134.58(16) N (1)#1-Zn (1)-O (11) 117.26(16) O (1)-Zn (1)-N (2)#2 111.74(16) N (1)#1-Zn (1)-N (2)#2 101.94(16) O (11)-Zn (1)-N (2)#2 91.26(16) O (1)-Zn (1)-O (10) 94.00(15) N (1)#1-Zn (1)-O (10) 92.46(15) O (11)-Zn (1)-O (10) 57.30(14) N (2)#2-Zn (1)-O (10) 148.52(15) O (13)-Zn (2)-O (6) 128.19(17) O (13)-Zn (2)-N (3)#1 106.79(18) O (6)-Zn (2)-N (3)#1 99.00(17) O (13)-Zn (2)-N (4)#3 97.81(16) O (6)-Zn (2)-N (4)#3 116.47(16) N (3)#1-Zn (2)-N (4)#3 107.23(18) Complex 4 Cd (1)-N (2)#1 0.225 4(3) Cd (1)-O (2) 0.226 0(3) Cd (1)-O (6) 0.229 3(3) Cd (1)-N (1)#2 0.232 4(3) Cd (1)-O (1W) 0.238 5(3) Cd (1)-O (1) 0.247 6(3) N (2)#1-Cd (1)-O (2) 150.06(11) N (2)#1-Cd (1)-O (6) 99.20(12) 0(2)-Cd (1)-O (6) 86.53(11) N (2)#1-Cd (1)-N (1)#2 110.59(12) O (2)-Cd (1)-N (1)#2 98.78(11) O (6)-Cd (1)-N (1)#2 89.27(11) N (2)#1-Cd (1)-O (1W) 84.09(11) O (2)-Cd (1)-O (1W) 92.60(10) O (6)-Cd (1)-O (1W) 174.82(9) N (1)#2-Cd (1)-O (1W) 84.09(11) N (2)#1-Cd (1)-O (1) 95.00(10) O (2)-Cd (1)-O (1) 55.21(9) O (6)-Cd (1)-O (1) 92.92(10) N (1)#2-Cd (1)-O (1) 153.64(11) O (1W)-Cd (1)-O (1) 90.77(10) Symmetry codes: #1:-x+1, y+1/2, -z+3/2, #2 x-1, -y+1/2, z+1/2, #3-x+1, y-1/2, -z+3/2, #4 x+1, -y+1/2, z-1/2 for 1; #1 x-1, y, z, #2-x-1, y+1, -z-1/2 for 2; #1 x-1, y, z, #2 x, -y+1/2, z+1/2, #3 x, -y-3/2, z+1/2 for 3; #1 x-1, y+1, z, #2 x, y, z+1 for 4. -

 扫一扫看文章
扫一扫看文章
计量
- PDF下载量: 0
- 文章访问数: 1362
- HTML全文浏览量: 166


 下载:
下载:







 下载:
下载:

