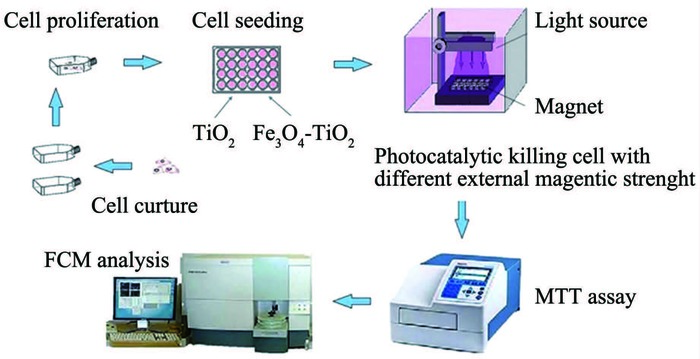
 Figure 1.
Schematic diagram of magnetic Fe3O4-TiO2 photocatalytic killing process and mechanism
Figure 1.
Schematic diagram of magnetic Fe3O4-TiO2 photocatalytic killing process and mechanism

纳米Fe3O4-TiO2靶向光动力疗法对肝癌细胞的杀伤效应
-
关键词:
- 光动力疗法
- / Fe3O4-TiO2纳米粒
- / 光催化
- / 机制
- / 肝癌细胞
English
Photocatalytic Killing Effect of Fe3O4-TiO2 Nanoparticles on Hepatoma Carcinoma Cells for Targeting Photodynamic Therapy
-
Key words:
- photodynamic therapy
- / Fe3O4-TiO2 nanoparticle
- / photocatalysis
- / mechanism
- / HepG2 cell
-
Worldwide statistical reports indicate that hepatic carcinoma, one of highest mortal malignancy, is the third leading cause of cancer mortality[1]. Although traditional treatment is feasible, it always accompa-nied with side effect, the systematic toxicity of drugs, undesirable treatment efficiency and substantial recurrence and mortality[2].
Photodynamic therapy (PDT), which based on the photochemical reactions of photosensitizer (PS)[3-4], is an effective, noninvasive, nontoxic therapeutics and has attracted wide attention and is applied to studies on cancer therapy[5]. The mechanism of PDT is that, the activated PS transfers its excited-state energy to nearby oxygen molecular to generate reactive oxygen species (ROS), such as OH·, H2O2 and HO2·, which could induce oxidative damage to the structure of various biological molecules through a series of peroxidative reactions and thus result in the death of cancer cells[6-11].
Classical photosensitizers are mainly from porphyrins and their derivatives[12]. However, their inherent limitations including unstable properties, high toxicity, and cumbersome synthesis procedure hinder their application of PDT. To overcome these weaknesses, titanium dioxide (TiO2), absorbing ultraviolet (UV) light to catalyze the generation of ROS[13-14], can be used as PS. It has the advantages of non-toxic, stability, easy synthesis. Unfortunately, TiO2 still has some drawbacks in the clinical use, such as insufficient selectivity, poor solubility and low efficiency resulting from the deficiency of cell-specific accumulation of TiO2 on cancer cells. Hence, an efficient TiO2 modification for higher selectivity and killing efficiency is needed[15-16].
Aiming at the disadvantages of TiO2, numerous platforms for targeting delivery have been studied, of which, magnetic nanoparticles have attracted extensive interest because of their target ability steered by an external magnetic field[17-18]. Therefore, a rational design to modify TiO2 with magnetic nanoparticles is fundamentally developed.
Magnetic Fe3O4 nanoparticles (NPs), as promising material because of alluring properties of chemical stability, magnetic properties and biocompatibility[19-20], have myriads of potential applications such as magnetic resonance imaging, catalysis, and drug delivery[21-27]. Moreover, due to the small particle size of magnetic NPs (10~100 nm), they can penetrate blood capillaries and reach the depths of target tumor tissue avoiding phagocytosis of reticuloendothelial system[28]. Thus magnetic NPs have a longer blood circulation and interaction time than common drugs. Therefore, magnetic Fe3O4-TiO2 NPs were supposed to possess various advantages including targeting action, high selectivity and efficiency, low side effect[29-30] in anticancer activities.
In addition, a synergistic anti-cancer effect could be obtained by the combination of magnetic NPs with PSs as drug or drug delivery system. The double materials translated into double functionality: generation of ROS under excitation light source and heat production under alternating magnetic field stimulation, coupling PDT to magnetic hyperthermia (MHT)[31]. Therefore, magnetic Fe3O4-TiO2 NPs as PS have a wide prospect in MHT and PDT for cancer.
The photokilling effect of magnetic targeted Fe3O4-TiO2 NPs on hepatoma carcinoma cells (HepG2) in different external magnetic fields was investigated under different light sources. The core-shell structure Fe3O4-TiO2 NPs applied in this study were reported in our previous study[32]. In this paper, the influence of different light sources, irradiation time, magnetic field strength and the concentration of Fe3O4-TiO2 NPs on the photocatalytic killing effect were investigated using HepG2 cells as a model. The changes of cell cycle, apoptosis rate and MMP of HepG2 cells treated by photocatalytic Fe3O4-TiO2 NPs in external magnetic field were detected by FCM, and the mechanism was explored through the mode of action between photo excited Fe3O4-TiO2 NPs and HepG2 cells. The results will provide a new idea for cancer therapy and theoretical basis for further clinical application.
1 Experimental
1.1 Instruments
Digital pH-meter (pH S-3, Shanghai Leici Device Works, China). CO2 incubator (Asheville NC Company, USA). Ultraclean workbench (GB-Ⅱ, Beijing New Technology Application Research Institute). Low-speed centrifuge (LDZ5-2, Beijing Medical Centrifuge Plant). Electric steam pressure sterilizers (LDZX-40, Shanghai Shenan Medical Instrument Factory). Intensity meter (VLX-3W, SP Company, USA). High pressure mercury lamp (GGZ, Shanghai Yaming Lighting Company). LED light (LX-1, Shenzhen Green Xing Lighting Technology Company). Flow cytometry instrument (FACS-Calibur, BD company, USA). Microplate reader (Bio-Rad Novapathtm, USA). Inverted microscope (Olympus Company, Japan). NdFeb Magnet (N10, N27, N40, China).
1.2 Materials
Fe3O4-TiO2 NPs were prepared and characterized early by research group[32], which displayed good dispersion, high photocatalytic activity, and excellent magnetic responsivity. The average particle size was 50 nm and the photoresponsive range of TiO2 was extended to 444 nm.
Human hepatoma carcinoma cell (Shanghai Institute of Cell Biology, Chinese Academy of Sciences, Shanghai, China). Dulbecco's modified Eagles medium (DMEM, Neuronbc Laboratories Company). 10%(V/V) fetal bovine serum (FBS, Yuanhengjinma Bio-technology Development Company, Beijing). 1% (V/V) Penicillin-streptomycin solution (100×, Beijing Solarbio Science & Technology Company). Trypsin-EDTA (0.25%, Beijing Solarbio Science & Technology Company). Dimethyl sulfoxide (DMSO, Beijing Solarbio Science & Technology Company). Thiazolyl blue (MTT, Beijing Solarbio Science & Technology Company). Rhodamine 123 kit (Rh-123, Keygen Biotech, Nanjing, China). Propidium Iodide (PI, Sigma, USA). Fetal bovine serum (FBS, Gibco, USA). Milli-Q (Millipore, Bedford, MA, USA).
1.3 Cell culture and treatment
HepG2 cells were maintained in a 37℃, 5% carbon dioxide environment in Dulbecco's modified Eagle's medium supplemented with 10% (V/V) fetal bovine serum (FBS) and 1% (V/V) Penicillin-Streptomycin.
Fe3O4-TiO2 NPs and TiO2 were suspended by fresh medium to different concentrations of 100, 200, 300, 400 and 500 μg·mL-1. When HepG2 cells approached the exponential growth phase, cells were transferred and cultured in a 24-well plate and allowed to grow for 24 h at 37 ℃ in a 5% CO2 incubator. Then the medium was replaced with 1 mL of fresh culture medium containing sonicated Fe3O4-TiO2 NPs and TiO2 respectively. With different external magnetic strength, samples were irradiated by high pressure mercury lamp (dominant wavelength is 365 nm) or light emitting diode (LED, dominant wavelength is 420~425 nm) at room temperature. A band-pass filter was applied to obtain a light wavelength between 348~372 nm. The light intensity at the liquid surface was measured by a VLX-3W radiometer-photometer. The schematic diagram of Fe3O4-TiO2 NPs photocatalytic killing HepG2 cells was shown in Fig. 1. After treatment, HepG2 cells were incubated for 24 h to measure cell viability and 4 h to detect cell cycle and MMP.
1.4 Experimental grouping
In our study, the experiment was divided into six groups:
Group Ⅰ: negative control group, routinely cultured HepG2 cells without any treatment.
Group Ⅱ: positive control group, HepG2 cells cultured in different concentrations of TiO2 were similarly treated with non-irradiation control group, irradiation experimental group and irradiation experimental group under magnetic field.
Group Ⅲ: non-irradiation control group, HepG2 cells were treated with different concentrations of Fe3O4-TiO2 NPs in absence of light.
Group Ⅳ: irradiation control group, HepG2 cells were respectively irradiated by high pressure mercury lamp or LED lamp for 20, 30 and 40 minutes without any nanoparticles.
Group Ⅴ: irradiation experimental group, HepG2 cells cultured in different concentrations of Fe3O4-TiO2 NPs were similarly treated with irradiation control group.
Group Ⅵ: irradiation experimental group under magnetic field, HepG2 cells were treated with different concentrations of Fe3O4-TiO2 NPs and irradiated by high pressure mercury lamp or LED lamp under magnetic field.
1.5 Cell viability analysis
To assess cell viability, the modified MTT assay protocol was used[33]. MTT (3-(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyl tetrazolium bromide) was dissolved in phosphate buffer solution (PBS, pH 7.4) at 5 mg·mL-1 and filtered to be sterilized. 40 μL of stock MTT solution was added to each well and incubated at 37 ℃ for 4 h. Culture medium was removed and 600 μL of DMSO was added to each well and mixed thoroughly to dissolve the dark blue formazan crystals. After a few minutes to ensure that all crystals were dissolved at room temperature, the solution were extracted from each well and placed in the corresponding wells of 24-well plates. Optical density (OD) values were measured on a microplate reader at wavelength of 492 nm, taking the solution without MTT as control.
1.6 Cell cycle analysis
Cell cycle was assessed by flow cytometric analysis of ethanol-fixed cells stained with DNA-binding dye PI. Flow cytometry analysis was performed on a flow cytometer with Cell Quest Pro software. The light-scatter channels were set on linear gains and the fluorescence channels on a logarithmic scale. A minimum of 1×104 cells were analyzed in each condition with threshold setting adjusted so that the cell debris was excluded from the data acquisition. The grain density of cells was assessed by side scatter (SSC)[34] and the results would be presented with the DNA histograms.
1.7 Mitochondrial membrane potential analysis
Mitochondrial depolarization was determined using cell apoptosis rhodamine123 (Rh123) detection kit, a lipophilic cation susceptible to the changes in MMP. It has a property of entering mitochondria with membrane polarization and emitting flavo-green fluorescence with exciting of 488 nm excitation. If the mitochondrial potential is disturbed, the dye entering mitochondria will reduce, and the flavo-green fluorescence will drop[35]. The cells were stained with Rh123 as described by the manufacturer, and the flavo-green fluorescence was detected by FCM. The strength of fluorescence corresponds to the value of MMP.
1.8 Statistical analysis
Experiments were repeatedly conducted for three times. Values were reported as mean ±SD. Statistical comparisons were made by t-test to detect significant differences using SPSS 17.0 software. P < 0.05 was considered to be statistically significant.
2 Results and discussion
2.1 Viability of HepG2 cells
2.2 Mechanism of photocatalytic killing
2.1.2 Influence of irradiation time
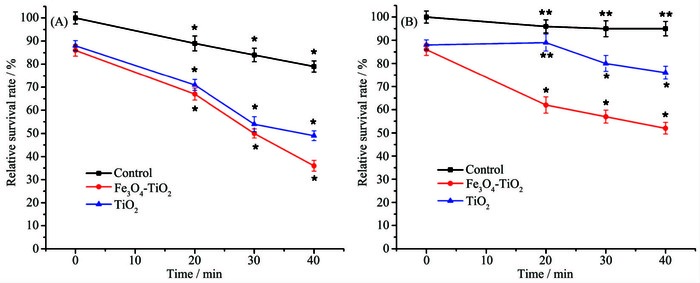
The effect of different ultraviolet and visible light irradiation time on the relative survival rate of HepG2 cells is presented in Fig. 3. With the extension of illumination time, the relative survival rate of HepG2 cells in each group markedly decreased (*P < 0.05). The relative survival rate of HepG2 cells in TiO2 group was greater than Fe3O4-TiO2 group at different time points.
Compared with visible light, for control and TiO2 groups, ultraviolet light had a more significant effect on photokilling efficiency, which was consistent with the fact that ultraviolet light is necessary for TiO2 photokilling cancer cells. However, in Fe3O4-TiO2 group, no significant difference was found in the relative survival rate of HepG2 cells between visible and ultraviolet light. It is suggested that Fe3O4 modified TiO2 made its light response range expanding from ultraviolet to visible region[32].
Taken together, Fe3O4-TiO2 killed HepG2 cells in a time-dependent manner. Fe3O4-TiO2 had many advantages over TiO2 on killing cancer cells, especially high photokilling efficiency under visible light[32].
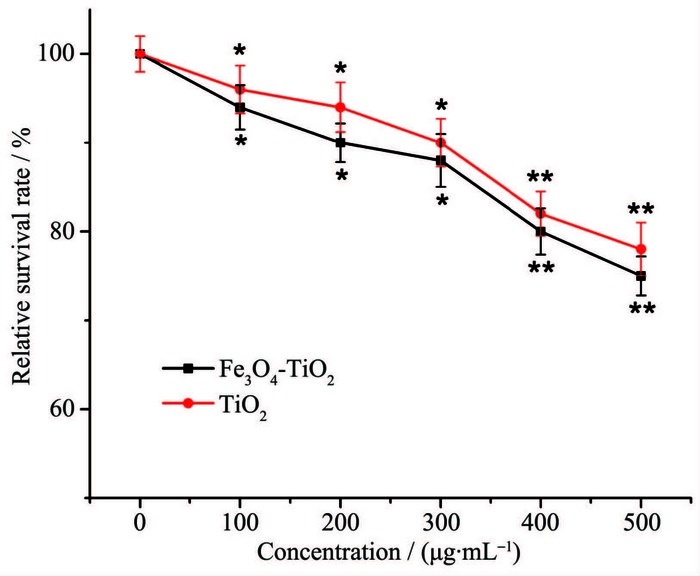
2.1.1 Biosafety assessment
Biosafety assessment was vital to evaluate the potential application of Fe3O4-TiO2 NPs in clinics. Here, modified MTT assay was performed to detect the toxicity of Fe3O4-TiO2 NPs (Fig. 2). The survival rate of HepG2 cells gradually decreased as the concentration of TiO2 and Fe3O4-TiO2 increasing, and the cytotoxicity of Fe3O4-TiO2 was slightly stronger than TiO2 without irradiation. When their concentration was greater than 400 μg·mL-1, a significant difference (**P < 0.05) existed, which was different from literature that nano-TiO2 was non-toxic to animals[36-37]. The possible reason is that at a high concentration and after long time, the tiny particle size of TiO2 and Fe3O4-TiO2 NPs interacting with HepG2 cells might damage the cell membrane or enter the HepG2 cells to cause necrosis[38-39].
However, when the concentrations of TiO2 and Fe3O4-TiO2 suspension were in the range of 0~300 μg·mL-1, there is no significant difference (**P > 0.05). Since it is widely accepted that the drugs used for the photocatalytic killing cancer cells should be non-toxic without irradiation, and thus TiO2 and Fe3O4-TiO2 NPs can be used as drugs for photocatalytic killing HepG2 cells.
2.1.4 Morphology
The cell morphology of Group Ⅰ, Ⅳ, Ⅴ and Ⅵ were observed by inverted microscope. The results are shown in Fig. 5. Normal HepG2 cells of Group Ⅰ adherently grew and the outlines were clear with the form of spindle or polygons. The microscopic structure could be observed, and individual cell shrank into round shape during recession phase (Fig. 5a). It was observed that HepG2 cells of Group Ⅳ, Ⅴ and Ⅵ tended to exhibit different morphological changes. Under the visible light irradiation, there was no significant change in cellular morphology (Fig. 5b). In contrast, cells irradiated by ultraviolet light markedly decreased, and parts of cells adherently grew, with others contracting into round shape and floated in the culture medium (Fig. 5c). Cellular numbers of Group Ⅴ were all reduced. Cellular debris was generated after ultraviolet light irradiation (Fig. 5e). Under visible light irradiation, cells shrank and the cell membrane became rough, indicating that cell apoptosis might occur (Fig. 5d). Compared with Group Ⅴ, the cellular number of Group Ⅵ had similar trend. Cells shrank into round shape and floated in the culture medium after ultraviolet light irradiation (Fig. 5f). After visible light irradiation, cells shrank and pseudopodia protruded, indicating that the killing effect of Fe3O4-TiO2 NPs under magnetic field might have two forms, including necrocytosis and cell apoptosis (Fig. 5g).
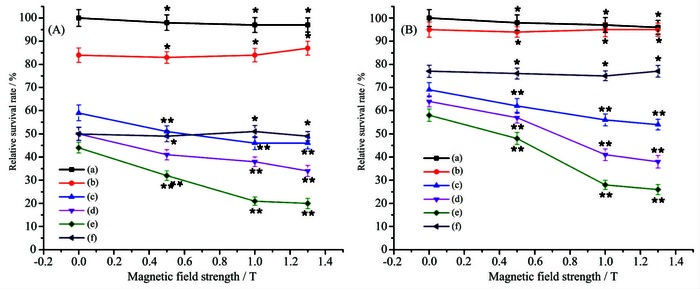
2.1.3 Influence of magnetic field strength and concentration
The effect of magnetic field strength and concentration of Fe3O4-TiO2 on the photocatalytic killing efficiency are shown in Fig. 4. With magnetic field strength ranging from 0 to 1.5 T, the relative survival rate of HepG2 cells in each group decreased in varying degrees, and Fe3O4-TiO2 group varied most and its killing efficiency is the strongest (**P > 0.05). It is suggested that magnetic Fe3O4-TiO2 under magnetic field could be targeted to kill cancer cells, thereby possessing high killing efficiency. To be specific, the killing effect of Fe3O4-TiO2 on HepG2 cells was markedly enhanced with the increased magnetic field strength in the range of 0~1.0 T. It has been reported that the photokilling effect of TiO2 NPs could be prominently improved when they entered cancer cells, especially the nucleus[40-41]. It is presumed that under external magnetic field, more nanoparticles would deposit onto the bottom of the culture plate, get in touch with the adherent growth HepG2 cells and enter the interior of HepG2 cells to kill cells. However, the trend of curve decreased slightly in the range of 1.0~1.5 T, which suggested that almost all of nanoparticles had deposited onto the bottom and the killing effect had reached its maximum limits.
The killing efficiency of Fe3O4-TiO2 increased with the increasing of the concentration of Fe3O4-TiO2. Therefore, the killing effect was speculated to be concentration-dependent. And when the concentration of suspension was 300 μg·mL-1, the view that killing efficiency of Fe3O4-TiO2 was much greater than TiO2 has been further confirmed.
From Fig. 4A and Fig. 4B, ultraviolet irradiation had more significant effect than visible light on the relative survival rate of HepG2 cells in control and TiO2 group, which can be speculated that visible light played a tiny role. However, in both cases, Fe3O4-TiO2 had showed strong killing effect. This further proved that its light response range was broadened to visible region (444 nm), lest tissues burned and mutations exposed to ultraviolet light.
The results suggested that killing effect of Fe3O4-TiO2 NPs on HepG2 cells could be enhanced under external magnetic field. Thus, the good magnetic responsivity of nanoparticles might be expected to be a drug for targeted therapy of cancer. It could reach the targeted tissue guided by external magnetic field, reduce damage to normal cells and enhance the killing effect.
2.2.2 MMP analysis
Mitochondrion is one of vital cell organs in the process of transmitting apoptosis signs, and depola-rization of MMP is the earliest event in the cascade reaction of apoptosis process[49]. Therefore, study on the changes of MMP plays an important role in clarifying mechanism caused by photocatalytic Fe3O4-TiO2 NPs. The changes on MMP of HepG2 cells caused by photocatalytic Fe3O4-TiO2 NPs in external magnetic field was presented in Fig. 7.
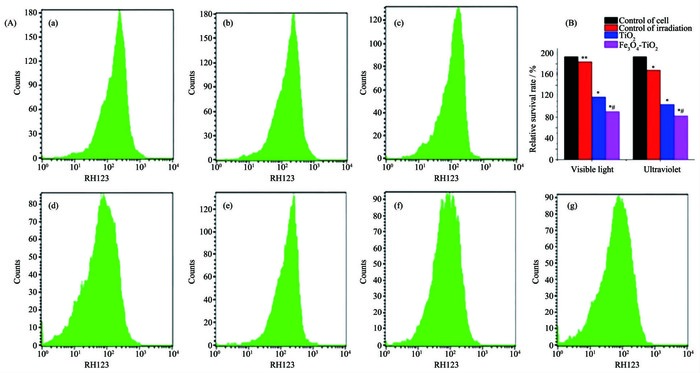
Rhodamine (Rh123) was used as a fluorescent probe for assessing the damage to mitochondria. Compared with other groups, Fe3O4-TiO2 group apparently increased the fluorescence of Rh123 on HepG2 cells (Fig. 7A). This phenomenon indicated a loss of mitochondrial function.
To find whether Fe3O4-TiO2 NPs can induce mitochondrial depolarization in HepG2 cells, fluore-scence intensity was quantified (Fig. 7B). Compared with cell control group, the MMP of HepG2 cells in visible light control group had no obvious change (**P > 0.05), but a marked decrease was found (*P < 0.05) in ultraviolet light control group, TiO2 group and Fe3O4-TiO2 group. Fe3O4-TiO2 NPs excited either by ultraviolet or visible light showed no obvious difference on the depolarization of MMP (#P > 0.05), while more significant impact than that of TiO2 was observed in both cases. The results further indicated that the mechanism of photocatalytic killing HepG2 cells was similar for two excitation conditions.
Mitochondria contributed to the production of ROS that is necessary for redox-dependent cellular processes. Mitochondria were carried out aerobic respiration. Excited Fe3O4-TiO2 produced a large amount of superoxide in cells. Surrounded by a high concentration of ROS, mitochondrial were attacked and its functional activity was restrained by cumulative ROS. As a result, cells were approaching to apoptosis because of lack of energy, and consequently HepG2 cell viability inevitably dropped.
Besides, to maintain mitochondrial function, normal MMP is necessary for a prerequisite in the maintenance of mitochondrial oxidative phosphorylation and the synthesis of adenosine triphosphate (ATP)[50]. The depolarization of MMP can cause a series of biochemical changes both inside and outside on mitochondrial membrane, including release of cytochrome C with a dual function of regulating the energy metabolism and apoptosis, altering cell membrane permeability, stopping the synthesis of ATP and releasing apoptosis inducing factor (AIF). Consequently, due to disruption of the cell membrane, the cascade reaction of apoptosis process will be induced and the cells will enter a nonreversible apoptosis process[51].
Mitochondrion is the control center of apoptosis[52]. In the apoptosis of mitochondrial regulation, reduction of MMP is an important indicator of the permeability and structural damage to mitochondrial membrane. Accordingly, in our experimental conditions, the photocatalytic Fe3O4-TiO2 NPs might kill HepG2 cells via mitochondrial pathway to induce apoptosis.
2.2.3 Mode of action
Apoptosis of HepG2 cells might be induced by photocatalytic Fe3O4-TiO2 NPs in two ways. One way is that ROS produced by photocatalytic Fe3O4-TiO2 NPs causes imbalance in intracellular redox state and membrane damages of HepG2 cells, so the perme-ability of the mitochondrial membrane changes and its MMP reduces, inducing HepG2 cell apoptosis[36]. Ano-ther way is that Fe3O4-TiO2 NPs enter intracellular of HepG2 cells by endocytosis or passive diffusion, dire-ctly damage mitochondria and other intracellular com-ponents, and induce apoptosis through ROS or photo-generated hole-electron pair upon their surface[38-40].
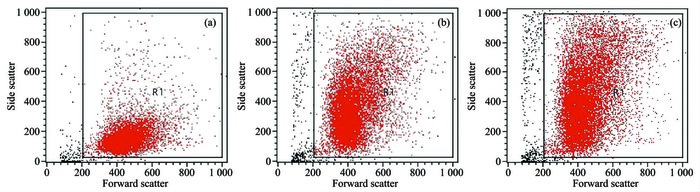
SSC parameters (Fig. 8) obtained by FCM can be used to judge whether Fe3O4-TiO2 enters HepG2 cells. The abscissa and ordinate represented FFC and SSC parameters of HepG2 cells, respectively. The quadrants were divided by solid line frame to distinguish between living and dead cells, where red and black dots stood for living and dead cells (or cell debris), respectively. Most of living cells located below quadrant in negative control group, and SSC parameters were small. While cell populations in Fe3O4-TiO2 and TiO2 groups shifted up and the SSC parameters increased, especially those in Fe3O4-TiO2 group. The result indicated that more Fe3O4-TiO2 NPs entered HepG2 cells than TiO2. It allowed conclusion that photocatalytic killing effect of Fe3O4-TiO2 NPs was stronger than TiO2 in external magnetic field.
According to the above experimental results, photocatalytic Fe3O4-TiO2 NPs could induce apoptosis of HepG2 cells through the second way. Photocatalytic Fe3O4-TiO2 NPs induced apoptosis through directly oxidative damage to mitochondria and other intracellular components.
2.2.1 Cell cycle analysis
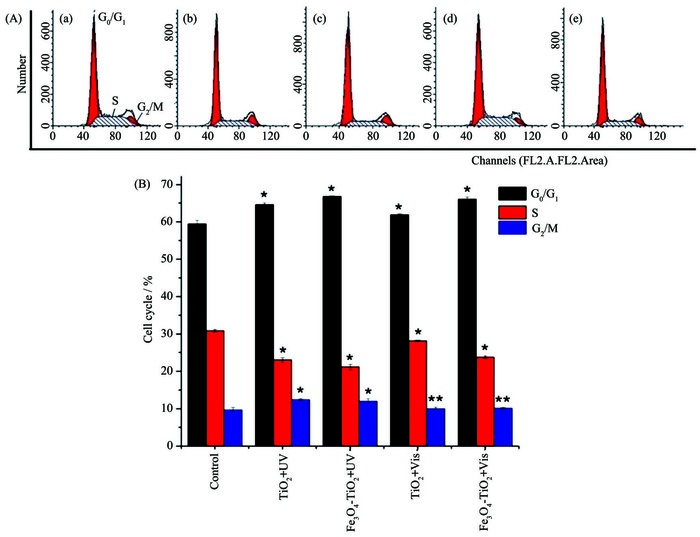
In external magnetic fields, the effect of Fe3O4-TiO2 NPs excited by ultraviolet and visible light on the cell cycle of HepG2 cells was examined by propidium iodide (PI) staining with FCM. The DNA histograms and the percentage of cell population in each phase are presented in Fig. 6.
Cell cycle analysis indicated decreased S phase and increased G0/G1, G2/M phase proportion, as well as apparent blockage of G0/G1 phase in response to TiO2 and Fe3O4-TiO2 NPs treatment (*P < 0.05), regar-dless of the irradiation conditions. The retardative effect and cell apoptosis rate of Fe3O4-TiO2 NPs were greater than TiO2. In the group of Fe3O4-TiO2, there were no significant differences in cell cycle arrest and apoptosis rate between ultraviolet and visible light (**P > 0.05), indicating that the mechanism might be similar in both cases.
Many studies have reported that the most characteristic effect of radiation (such as UV, X-ray and γ-ray, etc.) on eukaryotic cell cycle is arrested in G2/M phase[42-43]. Moreover, our previous study on the effect of photocatalytic nano-TiO2 on killing gastric cancer cells, demonstrated that the cell cycle was arrested in G2/M phase after ultraviolet irradiation alone for 20 min[44]. Therefore, it is supposed that the arrest of cell cycle in G2/M phase was caused by ultraviolet irradiation. The increase of cell numbers in G0/G1 phase and the decrease in S phase were caused by TiO2 and Fe3O4-TiO2 NPs. TiO2 and Fe3O4-TiO2 NPs driven by visible light didn't cause the arrest in G2/M phase, which also confirmed this hypothesis.
The arrest in G0/G1 phase is closely related to cellular proliferation and apoptosis. In G1 phase, mRNA, tRNA, ribosome and protein are mainly synthesized, which can provide necessary precursors for DNA synthesis in S phase[45]. Photocatalytic Fe3O4-TiO2 NPs could arrest HepG2 cells in G0/G1 phase and inhibit the transition to S phase. The reactive oxygen species (ROS)[46] generated by photocatalytic Fe3O4-TiO2 NPs might cause the direct damage to RNA and other substances synthesized in G1 phase. Meanwhile, the decrease of cell numbers in S phase indicated that the reduction or inhibition of DNA synthesis in S phase might be a way of Fe3O4-TiO2 NPs killing HepG2 cells.
It is reported that different cell cycle regulatory factors, proliferating cell nuclear antigen and p53 promote the transition of different cell cycle stages. Cyclin E-Cdk2 complex is the key regulatory factor in the cycle transition from G1 to S phase, whose activity is affected by related gene and protein[47]. In our experiment, HepG2 cells were arrested in G0/G1 phase by photocatalytic Fe3O4-TiO2 NPs, so it is speculated that the complex signals might be integrated and delivered by these genes and proteins, which would determine the next development of HepG2 cells such as entering into mitosis process or dormant G0 phase, causing programmed cell death or conducting self-repair[47-48]. The reason for cell cycle arrested in G0/G1 can be further pursued by investigating the changes of related genes and proteins.
3 Conclusions
Photocatalytic activities of magnetic Fe3O4-TiO2 NPs were investigated in HepG2 cells. Compared with TiO2, the core-shell structure Fe3O4-TiO2 NPs have magnetic targeted function, extend light response range to visible region and enhance photokilling efficiency. Moreover, the mechanism of photocatalytic Fe3O4-TiO2 NPs killing cancer cells was explored. Entering intracellular of HepG2, Fe3O4-TiO2 NPs stimulated the generation of ROS or photogenerated hole-electron pair, which directly damage mitoch-ondria and other intracellular components. Fe3O4-TiO2 induced apoptosis, mediated by arresting cell cycle in G0/G1 phase, reducing mitochondrial membrane potential, and mitochondrial depolarization. It can be concluded that Fe3O4-TiO2 magnetic NPs are of great value as effective drug in targeting photodynamic therapy. The potential of magnetic Fe3O4-TiO2 in MHT and PDT for cancer could also be expected.
-
-
[1]
Li H N, Cui Y N, Liu J, et al. J. Mater. Chem. B, 2014, 2: 3500-3510 doi: 10.1039/C4TB00321G
-
[2]
Jia Z Z, Tian F, Jiang G M. Curr. Ther. Res. Clin. Exp., 2013, 74:41-43 doi: 10.1016/j.curtheres.2012.12.006
-
[3]
Hou Z Y, Zhang Y X, Deng K R, et al. ACS Nano, 2015, 9 (3):2584-2599 doi: 10.1021/nn506107c
-
[4]
Master A, Livingston M, Gupta A S. J. Controlled Release, 2013, 168(1):88-102 doi: 10.1016/j.jconrel.2013.02.020
-
[5]
Chen Z L, Sun Y, Huang P, et al. Nanoscale Res. Lett., 2009, 4(5):400-408 doi: 10.1007/s11671-009-9254-5
-
[6]
Levy J, Obochi M. Photochem. Photobiol., 1996, 64(5):737-739 doi: 10.1111/php.1996.64.issue-5
-
[7]
Konan Y N, Gurny R, Allémann E. J. Photochem. Photobiol. B, 2002, 66(2):89-106 doi: 10.1016/S1011-1344(01)00267-6
-
[8]
Dougherty T J. Photochem. Photobiol., 1987, 45(6):879-889
-
[9]
Roy I, Ohulchanskyy T Y, Pudavar H E, et al. J. Am. Chem. Soc., 2003, 125(26):7860-7865 doi: 10.1021/ja0343095
-
[10]
Dodd N J F, Jha A N. Mutat. Res. Fundam. Mol. Mech. Mutagen., 2009, 660(1/2):79-82
-
[11]
Wang J, Guo Y W, Liu B, et al. J. Lumin., 2011, 131(2):231-237
-
[12]
Neghabi M, Zadsar M, Ghorashi S M B. Mater. Sci. Semicond. Process., 2014, 17:13-20 doi: 10.1016/j.mssp.2013.08.002
-
[13]
Cai R X, Kubota Y, Shuin T, et al. Cancer Res., 1992, 52(8): 2346-2348
-
[14]
Rozhkova E A, Ulasov I, Lai B, et al. Nano Lett., 2009, 9(9): 3337-3342 doi: 10.1021/nl901610f
-
[15]
Wang S Z, Gao R M, Zhou F M, et al. J. Mater. Chem., 2004, 14(4):487-493 doi: 10.1039/b311429e
-
[16]
Gao D, Agayan R R, Xu H, et al. Nano Lett., 2006, 6(11): 2383-2386 doi: 10.1021/nl0617179
-
[17]
Jain T K, Roy I, De T K, et al. J. Am. Chem. Soc., 1998, 120 (43):11092-11095 doi: 10.1021/ja973849x
-
[18]
Jain T K, Reddy M K, Morales M A, et al. Mol. Pharmaceu-tics, 2008, 5(2):316-327 doi: 10.1021/mp7001285
-
[19]
Weissleder R, Cheng H C, Bogdanova A, et al. J. Magn. Reson. Imaging, 1997, 7(1):258-263 doi: 10.1002/(ISSN)1522-2586
-
[20]
Saviuc C, Grumezescu A M, Holban A, et al. Biointerface Res. Appl. Chem., 2011, 1(2):64-71
-
[21]
Mahmoudi M, Hosseinkhani H, Hosseinkhani M, et al. Chem. Rev., 2011, 111(2):253-280 doi: 10.1021/cr1001832
-
[22]
Ho D, Sun X, Sun S. Acc. Chem. Res., 2011, 44(10):875-882 doi: 10.1021/ar200090c
-
[23]
Chen Y, Song B H, Lu L, et al. Nanoscale, 2013, 5:6797-6803 doi: 10.1039/c3nr01826a
-
[24]
Liang X Z. Mater. Lett., 2014, 137:447-449 doi: 10.1016/j.matlet.2014.09.083
-
[25]
Zhou L, He B Z, Huang J C. ACS Appl. Mater. Interfaces, 2013, 5(17):8678-8685 doi: 10.1021/am402334f
-
[26]
Rocher V, Siaugue J M, Cabuil V. Water Res., 2008, 42(4/ 5):1290-1298
-
[27]
Zhao F Y, Zhang B L, Feng L Y. Mater. Lett., 2012, 68:112-114 doi: 10.1016/j.matlet.2011.09.116
-
[28]
Zhang Y, Kohler N, Zhang M Q. Biomaterials, 2002, 23(7): 1553-1561 doi: 10.1016/S0142-9612(01)00267-8
-
[29]
Mahmoudi M, Sant S, Wang B, et al. Adv. Drug Delivery Rev., 2011, 63(1/2):24-46
-
[30]
Alexiou C, Schmid R J, Jurgons R, et al. Eur. Biophys. J., 2006, 35(5):446-450 doi: 10.1007/s00249-006-0042-1
-
[31]
Corato R D, Béalle G, Kolosnjaj-Tabi J, et al. ACS Nano, 2015, 9(3):2904-2916 doi: 10.1021/nn506949t
-
[32]
文雯, 高晓亚, 宋志英.物理化学学报, 2012, 28(9):2221-2230WEN Wen, GAO Xiao-Ya, SONG Zhi-Ying, et al. Acta Phys.-Chim. Sin., 2012, 28(9):2221-2230
-
[33]
Plumb J A. Methods Mol. Med., 2004, 88:165-169
-
[34]
Markovic Z M, Harhaji-Trajkovic L M, Todorovic-Markovic B M, et al. Biomaterials, 2011, 32(4):1121-1129 doi: 10.1016/j.biomaterials.2010.10.030
-
[35]
Scaduto R C, Grotyohann L W. Biophys. J., 1999, 76(1):469-477 doi: 10.1016/S0006-3495(99)77214-0
-
[36]
Bernard B K, Osheroff M R, Hofmann A, et al. J. Toxicol. Environ. Health, 1990, 29:417-429
-
[37]
Linnainmaa K, Kivipensas P, Vainio H. Toxicol. in Vitro, 1997, 11(4):329-335 doi: 10.1016/S0887-2333(97)00000-3
-
[38]
Thurn K T, Arora H, Paunesku T, et al. Nanomedicine, 2011, 7(2):123-130 doi: 10.1016/j.nano.2010.09.004
-
[39]
Lagopati N, Kitsiou P V, Kontos A I, et al. J. Photochem. Photobiol. A, 2010, 214(2/3):215-223
-
[40]
Zhu R R, Wang S L, Chao J. Mater. Sci. Eng. C, 2009, 29 (3):691-696 doi: 10.1016/j.msec.2008.12.023
-
[41]
Townley H E, Rapa E, Wakefield G, et al. Nanomedicine, 2011, 8(4):526-536
-
[42]
Maity A, Hwang A, Janss A, et al. Oncogene, 1996, 13(8): 1647-1657
-
[43]
Kao G D, McKenna W G, Maity A, et al. Cancer Res., 1997, 57(4):753-758
-
[44]
文雯, 高晓亚, 张爱平.应用化学, 2012, 28 (9):663-667WEN Wen, GAO Xiao-Yao, ZHANG Ai-Ping, et al. Chin. J. Appl. Chem., 2012, 28 (9):663-667
-
[45]
Wyllie A H. Brit. Med. Bull., 1997, 53(3):451-465 doi: 10.1093/oxfordjournals.bmb.a011623
-
[46]
Tang Y, Wang F, Jin C, et al. Environ. Toxicol. Phar., 2013, 36(1):66-72 doi: 10.1016/j.etap.2013.03.006
-
[47]
沈大棱, 吴超群.细胞生物学. Shanghai: Fudan University Press, 2006.SHEN Da-Leng, WU Chao-Qun. Cell Biology. Shanghai: Fudan University Press, 2006.
-
[48]
King K L, Cidlowski J A. Annu. Rev. Physiol., 1998, 60:601-617 doi: 10.1146/annurev.physiol.60.1.601
-
[49]
Green D R, Reed J C. Science, 1998, 281(5381):1309-1312 doi: 10.1126/science.281.5381.1309
-
[50]
Kroemer G, Zamzami N, Susin S A. Immunol. Today, 1997, 18(1):44-51 doi: 10.1016/S0167-5699(97)80014-X
-
[51]
Bemardi P, Scorrano L, Colonna R, et al. Eur. J. Biochem., 1999, 264(3):687-701 doi: 10.1046/j.1432-1327.1999.00725.x
-
[52]
Zoratti M, Szabo I. Biochim. Biophys. Acta, 1995, 1241(2): 139-176 doi: 10.1016/0304-4157(95)00003-A
-
[1]
-
Figure 4 Effect of Fe3O4-TiO2 NPs excited by ultraviolet (A) and visible light (B) on relative survival rate of HepG2 cells under external magnetic field
(a) Control of cell; (b) Control of visible light irradiation; (c) Fe3O4-TiO2 NPs (100 μg·mL-1); (d) Fe3O4-TiO2 NPs (200 μg·mL-1); (e) Fe3O4-TiO2 NPs (300 μg·mL-1); (f) TiO2 (300 μg·mL-1); Data were represented as mean ±SD (n=3);Remark: compared with the magnetic field strength of 1.0 T corresponding to each group, *P < 0.05, **P > 0.05
Figure 5 Morphological changes of HepG2 cells treated by different manners (×400)
(a) Normal HepG2 cells; (b) Visible light irradiation; (c) Ultraviolet irradiation; (d) Fe3O4-TiO2 NPs irradiated 30 min by visible light; (e) Fe3O4-TiO2 NPs irradiated 30 min by ultraviolet; (f) Fe3O4-TiO2 NPs irradiated 30 min by ultraviolet under external magnetic field; (g) Fe3O4-TiO2 NPs irradiated 30 min by visible light under external magnetic field; Scale bar=100 μm
Figure 6 Effect of photocatalysis Fe3O4-TiO2 NPs on cell cycle of HepG2 cells under external magnetic field
(a) Control of cell; (b) TiO2 excited by ultraviolet; (c) Fe3O4-TiO2 NPs excited by ultraviolet; (d) TiO2 excited by visible light; (e) Fe3O4-TiO2 NPs excited by visible light; Data were represented as mean ± SD (n=3); Remark: compared with the control of cell, *P < 0.05, **P > 0.05
Figure 7 Effect of photocatalytic Fe3O4-TiO2 NPs on MMP of HepG2 cells in external magnetic field
(a) Control of cell; (b) Control of visible light; (c) TiO2 excited by visible light; (d) Fe3O4-TiO2 NPs excited by visible light; (e) Control of ultraviolet; (f) TiO2 excited by ultraviolet; (g) Fe3O4-TiO2 NPs excited by ultraviolet; Data were represented as mean ±SD (n=3); Remark: compared with the control of cell, *P < 0.05, **P > 0.05; Comparison of the two Fe3O4-TiO2 NPs groups under irradiation of different light source, #P > 0.05
-

 扫一扫看文章
扫一扫看文章
计量
- PDF下载量: 0
- 文章访问数: 1166
- HTML全文浏览量: 136



 下载:
下载:







 下载:
下载:

