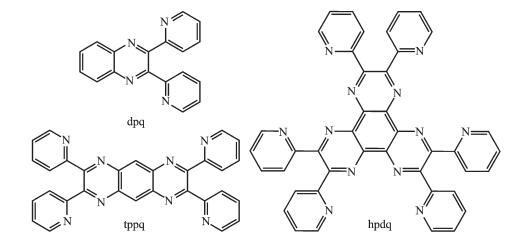
 Figure Scheme1.
Structure of three polypyridyl ligands
Figure Scheme1.
Structure of three polypyridyl ligands

多吡啶配体和银 (Ⅰ) 三明治配合物的构筑、结构及光谱学分析
English
Polypyridyl Ligands-Based Double-Decker Triggered by Silver (Ⅰ) Coordination: Crystal Structures and Spectroscopic Analysis
-
0 Introduction
With the aim to diversify the architecture and properties of metal organic coordination complexes, increasing attentions have been paid on the design of multidentate organic ligands[1-5], due to their versatile coordination modes. Among them N-donor heterocyclic multidentate ligands have been widely used in the construction of coordination complexes with intriguing topologies[6-10] and nanostructure[11-12], because of their versatile coordination behaviors.
Ag (Ⅰ) ion is one of the most widely used metal ion in the construction of coordination complex of nanostructure, since its d10 electron structure has no stronger energetic preferences for particular coordination geometry, which is benefit to the construction of versatile coordination complexes with intriguing structures[13-16], especially in combined with multidentate N-donor heterocyclic ligands[17-19]. However, the nanostructure based on Ag (Ⅰ) coordination complexes, as revealed in many literatures, depends greatly on the backbone of mutidentate ligands used. Meanwhile, the counter anions of silver salt also impact greatly the assembly structure in some cases.
Recently, a large polypyridyl ligand, 2, 3, 6, 7, 10, 11-hexakis (2-pyridyl) dipyrazino[2, 3-f:2′, 3′-h]-quinoxaline (hpdq) was reported to react with Ag (CF3SO3)[20], and the resultant complex comprises a novel double decker unit that is extended into three dimension architecture via the hydrogen bond interaction between CF3SO3- and the C-H bonds of hpdq. However, when reacting with Cd (Ⅱ) or Zn (Ⅱ) ions, hpdq exhibits distinctive coordination modes that facilitate to form a discrete planar structure based on single hpdq ligand[21]. Despite several intrinsic characteristics have been unveiled about the coordination interaction between Ag (Ⅰ) ion and polypyridyl quinoxaline ligands, some questions still remain unsettled about the structural dependence of double decker on the backbone of polypridyl quinoxaline ligands. Meanwhile, the influence of counter anions on the packing mode of double decker units is still not clear.
In order to promote solving these questions, the coordination interaction between AgNO3 and several polypyridyl quinoxaline ligands with different size and pyridine number (Scheme 1), i.e. 2, 3-di (pyridin-2-yl) quinoxaline (dpq), 2, 3, 7, 8-tetra (pyridin-2-yl) pyrazino[2, 3-g]quinoxaline (tppq) and 2, 3, 6, 7, 10, 11-hexakis (2-pyridyl) dipyrazino[2, 3-f:2′, 3′-h]quinoxaline (hpdq) had been studied. Based on these ligands, three coordination complexes [Ag (dpq)(NO3)2]n (1), [Ag4(tppq)2 (NO3)4] (2) and [Ag6(hpdq)2(NO3)6] (3) have been synthesized. Single crystal analyses reveal distinctive packing mode of double-decker unit for 1~3, associated with different double decker depending on the used ligands. The packing mode of double decker in 3 differs significantly from that reported in literature[20], due to different counter anions used. Furthermore, both fluorescence and UV-absorption spectra titration were utilized to investigate the assembly process of the organic ligands and Ag (Ⅰ) ion in solution. The most obvious spectral variation was observed for hpdq, indicative of potential application of hpdq as a molecular modle in sepctral detection of metal ions.
1 Experimental
1.1 Materials and method
All the starting materials for the synthesis were of reagent grade and used as received. All the solvents used for titration measurements were puried by standard procedures. Hpdq was synthesized according to literature[21]. Elemental analyses (C, H, and N) were performed on a Perkin-Elmer 240C analyzer. IR spectra in the 4 000~400 cm-1 range were measured on a TENSOR 27 OPUS FTIR spectrometer using KBr disks dispersed with sample powders. UV-Vis absorption spectra were measured with a Hitachi U-3010 UV-Vis spectrophotometer. Fluorescence spectra were recorded at room temperature on a Varian Cary Eclipse uorescence spectrometer.
1.2 Preparation of complex 1, 2 and 3
Complexes 1, 2 and 3 were obtained through the reaction of AgNO3 with respective polypyridyl ligands by solution diffusion method at room temperature.
Complex 1: A buffer layer of dichloromethane-acetonitrile (12 mL, 1:1, V/V) was carefully layered over a dichloromethane solution (2 mL) of dqp (0.1 mmol). Then a solution of AgNO3 (0.2 mmol) in acetonitrile (3 mL) was layered over the buffer layer. After ca. four weeks colourless block crystals were obtained with 38% yield (based on dbq). Anal. Calcd. for C18H12N6O6Ag2(%): C 34.64, H 1.94, N 13.47; Found (%): C 34.59, H 1.57, N 13.84. FTIR (KBr pellets, cm-1): 1 734w, 1 700w, 1 684m, 1 653m, 1 559s, 1 540m, 1 507w, 1 473m, 1 457m, 1 386s, 419w.
Complex 2: A buffer layer of dichloromethane-acetonitrile (10 mL, 1:1, V/V) was carefully layered over a dichloromethane solution (3 mL) of tppq (0.1 mmol). Then a solution of AgNO3 (0.3 mmol) in acetonitrile (3 mL) was layered over the buffer layer. Orange red block-shaped crystals were obtained after ca. five weeks in 30% yield (based on tppq). Anal. Calcd. for C15.25H10.25N5O3.375Cl0.5Ag (2·CH2Cl2·1.5H2O, %): C 41.33, H 2.33, N 15.80; Found (%): C 41.72, H 2.53, N 16.05. FTIR (KBr pellets, cm-1): 1 684m, 1 653m, 1 559m, 1 540w, 1 457m, 1 340s.
Complex 3: A buffer layer of chloroform-acetonitrile (12 mL, 1:1, V/V) was carefully layered over a chloroform solution (2 mL) of hpdq (0.1 mmol). Then a solution of AgNO3 (0.3 mmol) in acetonitrile (3 mL) was layered on the buffer layer. Yellow block-shaped crystals were obtained after ca. five weeks in 35% yield (based on hpdq). Anal. Calcd. for C96.5H60.5N30O18Cl37.5Ag6(3·12.5CHCl3, %): C 29.68, H 1.56, N 10.76; Found (%): C 31.82, H 2.63, N 13.15. FTIR (KBr pellets, cm-1): 1 734w, 1 700w, 1 684w, 1 653w, 1 559m, 1 540w, 1 507w, 1 362s, 419w.
Note: The crystal of complex 3 is not stable at room temperature, especially upon the easily losing of the guest solvent molecules (CHCl3) during its collection from solvents, which leads to the deviation of experimental Element. Anal. data from the calculated values.
1.3 X-ray data collection and structure determinations
Single-crystal X-ray diffraction measurements for 1 and 2 were carried out on a Rigaku SCX-mini diffractometer at 293(2) K with Mo Kα radiation (λ=0.071 073 nm) by ω scan mode. The structures were solved by direct methods using the SHELXS program of the SHELXTL package and refined with SHELXL[22] (semi-empirical absorption corrections were applied by using the SADABS program). The non-hydrogen atoms were located in successive dierence Fourier syntheses and refined using anisotropic thermal para-meters on F2. All hydrogen atoms of ligands were generated theoretically at the specific atoms and refined isotropically using fixed thermal factors. Single-crystal X-ray diffraction measurements for complex 3 was carried out on a Bruker Smart 1000 CCD diffractometer at 113 K with Mo Kα radiation (λ=0.071 073 nm). The structure was solved by direct methods with the SHELXS-97 program [23]. Refinements were done by full-matrix least-squares techniques on F2 with SHELXL-97[24]. The hydrogen atoms bound to carbon were located by geometrically calculations. All non-hydrogen atoms were refined by full-matrix least-squares techniques. The electron density of the disordered guest molecules in complex 2 and 3 was treated using the SQUEEZE routine of PLATON[25-27]. The results were appended to the bottom of the CIF file. Detailed crystallographic data of 1, 2 and 3 (after the SQUEEZE) are summarized in Table 1.
Complex 1 2 3 Empirical formula Cl8Hl2Ag2N6O6 C15H9AgN5O3 C84H48Ag6N30Ol8 Formula weight 624.08 4l5.l4 24l2.75 Crystal system Monoclinic Orthorhombic Trigonal Space group P2l/n Fddd P3lc a / nm l.072 8(2) l.382 l (3) 2.069 7(3) b/ nm 0.873 3(l7) l.805 2(4) 2.069 7(3) c / nm 2.ll4 7(4) 5.257 3(ll) l.904 9(4) α/(°) l0l.l5(3) V/ nm3 l.950 2(7) l3.ll7(5) 7.066(2) Z 2 32 2 Dc/ (g·cm-3) 2.l26 l.682 l.l34 μ/ mm-l 2.06l l.252 0.870 F(000) l 2l6 6 560 2 376 GOF l.068 l.096 l.ll6 Reflections collected l6303 24 848 4 l65 Unique reflections 3 448 2 90l 4 l65 Rint 0.036 2 0.06l 9 0.000 0 Rl [I>2σ(I))] 0.036 8 0.052 0 0.099 0 wR2(all data) 0.042 5 0.l34 3 0.294 8 CCDC: 868734, 1; 868735, 2; 790268, 3.
2 Results and discussion
2.1 Crystal structure analysis of complexes 1~3
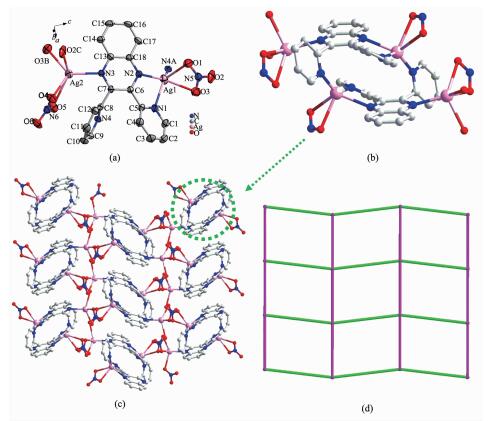
Complex 1 crystallizes in the P21/n space group with an asymmetric unit consisting of one dpq ligand and two crystallographically independent Ag (Ⅰ) ions (Fig. 1a). Ag1 ion is five coordinated by three N atoms (N1, N2, N4A) from two dpq ligands and two O atoms (O1 and O3) from one nitrate ion, while Ag2 ion is five coordinated by one N atom (N3) from dpq and four O atoms (O2C, O3B, O4 and O5) from three nitrate ions. All of the N-Ag-N angles (69.90(11)°~134.64(12)°), O-Ag-N angles (91.66(12)° to 125.23(11)°), Ag-N (0.222 4(3)~0.258 6(4) nm) and Ag-O (0.235 2(3)~0.258 6(4) nm) bond lengths are within the normal range[20, 26]. It is worth noticing that two dpq ligands are linked to form a double decker unit via N-Ag1 bonds (Fig. 1b). Furthermore, each double decker is linked with other four neighboring one through Ag2-NO3 coordination interaction, leading to a two-dimensional (2D) packing net (Fig. 1c). Simplifying the double decker unit as a four-connecting node generates a (4, 4) topologic structure for complex 1 (Fig. 1(d)).
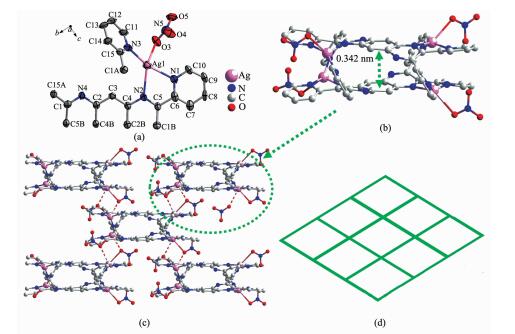
Complex 2 crystallizes in the Fddd space group, and the asymmetric unit consists of one quarter of a tppq ligand, one Ag (Ⅰ) ion and one nitrate ion, as shown in Fig. 2a. The Ag (Ⅰ) ion is four coordinated by three N atoms (N1, N2, N3) from tppq and one O atom (O3) from nitrate ion. Meanwhile, a double decker unit was also observed as in complex 2, but with four coordinated Ag (Ⅰ) ions (Fig. 2b). Inspection of the double decker in 2 reveals a centroid-to-centroid distance of 0.342 nm between the central benzene ring of two tppq ligands, indicative of π-π interaction existing between two ligands. Furthermore, each double decker unit is further linked with four neighboring units through a weak Ag-O coordination interaction (0.280 51 nm), leading to a 2D network (Fig. 2c). All of the N-Ag-N angles (68.63(14)°~152.08(16)°), O-Ag-N angles (90.43(16)°~130.40 (15)°), the bond length of Ag-N (0.226 8(5)~0.245 5(4) nm) and Ag-O (0.259 5(5) nm) bond length are within normal range[28]. Simplifying the double decker as a four-connecting node generates a (4, 4) topology structure for complex 2 (Fig. 2 (d)).
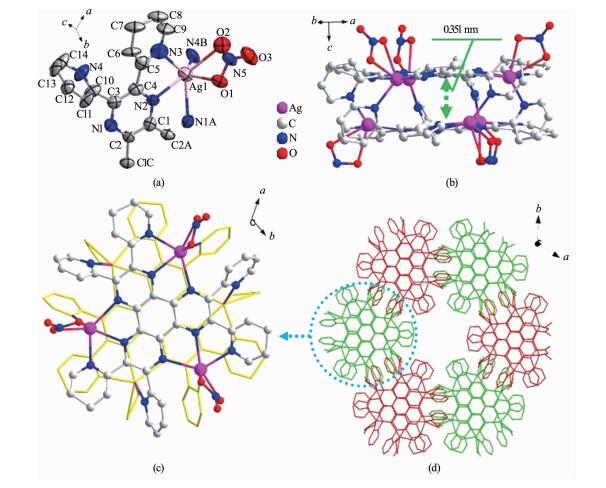
The asymmetric unit of 3 consists of one third of hpdq ligand and one Ag (Ⅰ) ion (Fig. 3a). Differing from the case in 1 and 2, the Ag (Ⅰ) ion in 3 is six coordinated by four N atoms (N1A, N2, N3, N4B) from hpdq and two O atoms (O1, O2) from a nitrate ion, leading to a deformed octahedral geometry. As shown in Fig. 3b and c, 3 assembled to 3D structure based on a similar double decker as in 1 and 2, but with six Ag (Ⅰ) ions. The planes of two ligands within one double decker are nearly parallel with each other, with a centroid-to-centroid distance of 0.351 nm between two central benzene rings, indicative of the presence of π-π interaction between the rings.
Despite similar structure of the double decker in 3 to that in literature[20], several vital differences were revealed via detailed inspection of crystal structure:
(1) The complex reported in literature[20] consists of two kinds of cyrstallographically independent Ag (Ⅰ) that are both five coordinated. Whereas the Ag (Ⅰ) ions in 3 is crystallographically equivalent and six coordinated. In addition, the distance between two hpdq ligands within one double decker of 3 (0.351 nm) is slight smaller than that reported in literature (0.364 nm), due probably to different coordination environment of Ag (Ⅰ) ions as well as different hydrogen bond pattern between two hpdq plane (Fig.S1).
(2) The most greatest difference lies in the packing pattern of double deckers. The double deckers in literature were linked with each other through the O…H-C and F…H-C hydrogen bonds between CF3SO3- anion and the C-H of hpdq, whereas that in 3 was completed only through O…H-C hydrogen bond (Fig.S2). The difference results in distinctive packing pattern of double decker (Fig.S3) and thereby different cell parameters and space group between 3 (a=2.069 7(3) nm, c=1.904 9(4) nm, V=7.066(2) nm3, P31c group) and the complex reported in literature (a=3.031 9(2) nm, c=1.011 9(1) nm, V=8.055 6(11) nm3, R3 group)[20]. Different packing mode of double decker in 3 from that of literature validates the vital role of counter anions in controlling the assembly architecture of Ag (Ⅰ)-based coordination complexes.
Detailed Comparison among 1~3 reveals some notable differences in their double decker units as well as the networks. Firstly, the Ag (Ⅰ) ions exhibit different coordination number in the double decker of 1, 2 and 3, that is 5 4 and 6, respectively. Secondly, the double deckers are linked with each other by strong O-Ag bond, weak O-Ag bond ( > 0.28 nm) and O…H-C hydrogen bond in 1, 2 and 3, respectively. These differences can be reasonably interpreted in terms of different backbone of used polypyridyl ligands. The strong O-Ag linkage between adjacent double deckers in 1 is repressed in 2 and 3, due probably to increasing steric hindrance of larger ligands, which therefore brings about elongated O-Ag linking in 2, and even more facilitates the hydrogen bond interaction between counter anions and Ag (Ⅰ) ions in 3. This underlines the important role of counter anions in the assembly of Ag (Ⅰ) ions and hpdq ligands.
2.2 Monitoring the coordination interaction by spectral titration
dpq, tppq and hpdq all are soluble in CH3CN and are luminescent owing to their conjugating system, which encourages us to investigate and compare their coordination interaction with Ag (Ⅰ) ions in solution by spectral analysis method.
All of polypyridine ligands were prepared with a concentration of 5×10-5 mol·L-1 using CH3CN as solvent, while AgNO3 solution (0.015 mol·L-1) was prepared using acetonitrile as solvent. During titration, the volume of the polypryridyl ligand solution (3 mL) can be regarded as constant because of negligible added volume of metal ions solution. For fluorescence spectra measurements, excitation and emission slit width were both set as 5 nm.
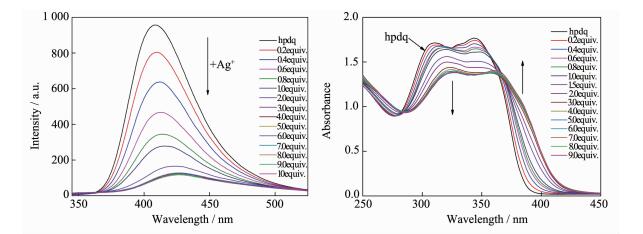
The Fluorescence variations of hpdq upon the addition of Ag (Ⅰ) ions were summarized in Fig. 4(a). At beginning of the addition of Ag (Ⅰ) ion, the strongest emission peak of hpdq at 409 nm was quenched sharply along with moderate red shift of emission wavelength. The variation becomes stagnant when the addition quantity of Ag (Ⅰ) ion approximately approaches to 4 equiv. of hpdq. Finally, the emission peak shifts to 427 nm with intensity less than 15% of the initial peak. The UV-Vis spectra of hpdq were collected in Fig. 4(b). The addition of Ag (Ⅰ) results in the sequential redshift of absorption peaks at 308 and 343 nm, along with the decreasing of the absorbance. The appearance of several isosbestic points between 360~375 nm at different molar ratio of Ag (Ⅰ) to ligands indicates the multistep assembly of hpdq ligand upon interacting with Ag (Ⅰ), which is consistent with the coordination of three Ag (Ⅰ) ions with one hpdq as observed in the crystal analysis of 3. On the other hand, the variation of Fluorescence and UV-Vis spectra almost has the same equilibrium point with a molar ratio of nhpdq :nAg (Ⅰ) equal to 1:3, which is consistent with the ratio of nhpdq:nAg (Ⅰ) observed in 3. In addition, the spectral variations of tppq and dpq upon the addition of Ag (Ⅰ) ion were also investigated (Fig.S4 and S5), and much more dull variations were observed in comparison with that of hpdq system, due probably to the less π-π interaction of ligands within the double decker in 1, 2 than in 3. This indicates that hpdq is a much sensitive spectral sensor for metal ion detection than tppq and dpq.
3 Conclusions
In summary, three polypyridyl ligands, dpq, tppq and hpdq, were investigated in their coordination interaction with Ag (Ⅰ) ion by single crystal analysis and spectral analysis. Single crystal structure analysis unveiled different double decker unit as well as different assembly framework of the resultant complexes 1~3, which was attributed to the co-operation of counter anions and different polypyridyl ligands. Meanwhile, Spectral titration was utilized to study the coordination interaction of polypyridyl ligands with Ag (Ⅰ) in solution, and it was observed that the spectral response of hpdq to the addition of Ag (Ⅰ) ions is much more sensitive than the other two ligands, which can be used to guide the design of new organic spectral sensor for Ag (Ⅰ) ions detection.
Supporting information is available at http://www.wjhxxb.cn
-
-
[1]
Ma L, Mihalcik D J, Lin W. J. Am. Chem. Soc., 2009, 131: 4610-4612 doi: 10.1021/ja809590n
-
[2]
Li Y P, Yang H R, Zhao Q, et al. Inorg. Chem., 2012, 51: 9642-9648 doi: 10.1021/ic300738e
-
[3]
Yang X L, Wu C D. Inorg. Chem., 2014, 53:4797-4799 doi: 10.1021/ic500531k
-
[4]
Zhang Y B, Furukawa H, Ko N, et al. J. Am. Chem. Soc., 2015, 137:2641-2650 doi: 10.1021/ja512311a
-
[5]
Xue D X, Cairns A J, Belmabkhout Y, et al. J. Am. Chem. Soc., 2013, 135:7660-7667 doi: 10.1021/ja401429x
-
[6]
Tian D, Chen Q, Li Y, et al. Angew. Chem. Int. Ed., 2014, 53:837-841 doi: 10.1002/anie.201307681
-
[7]
Wang Y T, Fan H H, Wang H Z, et al. Inorg. Chem., 2005, 44:4148-4150 doi: 10.1021/ic0504137
-
[8]
Harris K, Sun Q F, Sato S, et al. J. Am. Chem. Soc., 2013, 135:12497-12499 doi: 10.1021/ja4043609
-
[9]
Zhao Q, Liu X M, Song W C, et al. Dalton Trans., 2012, 41: 6683-6688 doi: 10.1039/c2dt00002d
-
[10]
Li X H, Chen Z, Zhao Q, et al. Inorg. Chem., 2007, 46:5518-5527 doi: 10.1021/ic062041n
-
[11]
Osuga T, Murase T, Fujita M. Angew. Chem. Int. Ed., 2012, 51:12199-12201 doi: 10.1002/anie.v51.49
-
[12]
Cui Y, Lee S J, Lin W. J. Am. Chem. Soc., 2003, 125:6014-6015 doi: 10.1021/ja029926s
-
[13]
Lubick N. Environ. Sci. Technol., 2008, 42:8617-8617 doi: 10.1021/es8026314
-
[14]
Hamasaki Y, Nakashima N, Niidome Y. J. Phys. Chem. C, 2013, 117:2521-2530 doi: 10.1021/jp306469s
-
[15]
Njua E Y, Steiner A, Stahl L. Inorg. Chem., 2010, 49:2163-2172 doi: 10.1021/ic9019537
-
[16]
Zhang X H, Zhao Q, Liu X M, et al. Talanta, 2013, 108:150-156 doi: 10.1016/j.talanta.2013.02.071
-
[17]
Nakamura T, Ube H, Shionoya M. Angew. Chem. Int. Ed., 2013, 52:12096-12100 doi: 10.1002/anie.201306510
-
[18]
Li X, Xu H, Kong F, et al. Angew. Chem. Int. Ed., 2013, 52: 13769-13773 doi: 10.1002/anie.201307650
-
[19]
Liu C S, Chen P Q, Yang E C, et al. Inorg. Chem., 2006, 45: 5812-5821 doi: 10.1021/ic060087a
-
[20]
Xiao Z Y, Zhao X, Jiang X K, et al. Chem. Mater., 2011, 23: 1505-1511 doi: 10.1021/cm103182e
-
[21]
Zhao Q, Li R F, Xing S K, et al. Inorg. Chem., 2011, 50: 10041-10046 doi: 10.1021/ic2008182
-
[22]
Sheldrick G M. SHELXTL Ver6. 1, Program for Solution and Refinement of Crystal Structures, University of Göttingen, Germany, 1998.
-
[23]
Sheldrick G M. SHELXS-97, Program for the Solution of Crystal Structures. University of Göttingen, Germany, 1997.
-
[24]
Sheldrick G M. SHELXL-97, Program for Crystal Structure Refinement. University of Göttingen, Germany, 1997.
-
[25]
Spek A L. PLATON, Utrecht University, Utrecht, The Netherlands, 2008.
-
[26]
Tan C H, Yang S H, Champness N R, et al. Chem. Commun., 2011, 47:4487-4489 doi: 10.1039/c1cc10378d
-
[27]
Yang W B, Greenaway A, Lin X, et al. J. Am. Chem. Soc., 2010, 132:14457-14469 doi: 10.1021/ja1042935
-
[28]
Zhang Z Y, Deng Z P, Huo L H, et al. Inorg. Chem., 2013, 52:5914-5923 doi: 10.1021/ic400055t
-
[1]
-
Figure 1 (a) Coordination geometry of Ag (Ⅰ) in 1; (b) The double-decker unit in 1; (c) The 2D network of 1; (d) Schematic representation of (4, 4)-connected topology of 1
Thermal ellipsoid at 50% probability; All hydrogen atoms are omitted for clarity; Symmetry codes: A:-x, 1-y, 2-z; B:-0.5+x, 0.5-y, 0.5+z; C:-x, y, 2-z
Figure 2 (a) The coordination geometry of Ag (Ⅰ) in 2; (b) The double-decker unit in 2; (c) The 2D network of 2 constructed by weak O-Ag bond (0.280 51 nm, in dotted bond); (d) Schematic representation of (4, 4)-connected topology of 2
Thermal ellipsoid at 50% probability; All hydrogen atoms are omitted for clarity; Symmetry codes: A: 1.25-x, 1.25-y, z; B: x, 1.25-y, 0.25-z
Figure 3 (a) The coordination geometry of Ag (Ⅰ) in 3; (b) View of the double-decker in 3 along the direction normal to c axis; (c) View of the double-decker in 3 from ab plane; (d) The packing diagram of double deckers
Thermal ellipsoid at 50% probability; All hydrogen atoms are omitted for clarity; Symmetry codes: A: 1-y, x-y z; B: 1-y, 1-x, 1.5-z; C:1-x+y, 1-x, z
Table 1. Crystal data and structure renement parameters for complexes 1~3
Complex 1 2 3 Empirical formula Cl8Hl2Ag2N6O6 C15H9AgN5O3 C84H48Ag6N30Ol8 Formula weight 624.08 4l5.l4 24l2.75 Crystal system Monoclinic Orthorhombic Trigonal Space group P2l/n Fddd P3lc a / nm l.072 8(2) l.382 l (3) 2.069 7(3) b/ nm 0.873 3(l7) l.805 2(4) 2.069 7(3) c / nm 2.ll4 7(4) 5.257 3(ll) l.904 9(4) α/(°) l0l.l5(3) V/ nm3 l.950 2(7) l3.ll7(5) 7.066(2) Z 2 32 2 Dc/ (g·cm-3) 2.l26 l.682 l.l34 μ/ mm-l 2.06l l.252 0.870 F(000) l 2l6 6 560 2 376 GOF l.068 l.096 l.ll6 Reflections collected l6303 24 848 4 l65 Unique reflections 3 448 2 90l 4 l65 Rint 0.036 2 0.06l 9 0.000 0 Rl [I>2σ(I))] 0.036 8 0.052 0 0.099 0 wR2(all data) 0.042 5 0.l34 3 0.294 8 -

 扫一扫看文章
扫一扫看文章
计量
- PDF下载量: 0
- 文章访问数: 1429
- HTML全文浏览量: 144


 下载:
下载:




 下载:
下载:

